I wrote this post at about the same time Germany won the World Cup in Rio de Janeiro in 2014. There’s been a lot of moving and shaking in the world of exogenous ketones since then, not to mention soccer. Looking back on my post, I still consider it relevant in terms of what exogenous ketones possibly can (and cannot) do for performance. In this case, to see if exogenous ketone esters provide me a “boost” by allowing me to do the same amount of work while expending less energy (and work at a relatively lower VO2) compared to no supplementation.
I’m getting an increasing number of questions about exogenous ketones. Are they good? Do they work for performance? Is there a dose-response curve? If I’m fasting, can I consume them without “breaking” the fast? Am I in ketosis if my liver isn’t producing ketones, but my BOHB is 1.5 mmol/L after ingesting ketones? Can they “ramp-up” ketogenesis? Are they a “smart drug?” What happens if someone has high levels of both glucose and ketones? Are some products better than others? Salts vs esters? BHB vs AcAc? Can taking exogenous ketones reduce endogenous production on a ketogenic diet? What’s the difference between racemic mixtures, D-form, and L-form? What’s your experience with MCTs and C8?
Caveat emptor: the following post doesn’t come close to answering most of these questions. I only document my experience with BHB salts (and a non-commercial version at that), but say little to nothing about my experience with BHB esters or AcAc esters. But it will provide you will some context and understanding about what exogenous ketones are, and what they might do for athletic performance. We’ll likely podcast about the questions and topics above and cover other aspects of exogenous ketones in more detail.
—P.A., June 2018
§
Original publication date: August 14, 2014
Last year I wrote a couple of posts on the nuances and complexities of ketosis, with an emphasis on nutritional ketosis (but some discussion of other states of ketosis—starvation ketosis and diabetic ketoacidosis, or DKA). To understand this post, you’ll want to at least be familiar with the ideas in those posts, which can be found here and here.
In the second of these posts I discuss the Delta G implications of the body using ketones (specifically, beta-hydroxybutyrate, or BHB, and acetoacetate, or AcAc) for ATP generation, instead of glucose and free fatty acid (FFA). At the time I wrote that post I was particularly (read: personally) interested in the Delta G arbitrage. Stated simply, per unit of carbon, utilization of BHB offers more ATP for the same amount of oxygen consumption (as corollary, generation of the same amount of ATP requires less oxygen consumption, when compared to glucose or FFA).
I also concluded that post by discussing the possibility of testing this (theoretical) idea in a real person, with the help of exogenous (i.e., synthetic) ketones. I have seen this effect in (unpublished) data in world class athletes not on a ketogenic diet who have supplemented with exogenous ketones (more on that, below). Case after case showed a small, but significant increase in sub-threshold performance (as an example, efforts longer than about 4 minutes all-out).
So I decided to find out for myself if ketones could, indeed, offer up the same amount of usable energy with less oxygen consumption. Some housekeeping issues before getting into it.
- This is a self-experiment, not real “data”—“N of 1” stuff is suggestive, but it prevents the use of nifty little things likes error bars and p-values. Please don’t over interpret these results. My reason for sharing this is to spark a discussion and hope that a more systematic and rigorous approach can be undertaken.
- All of the data I’ll present below were from an experiment I did with the help of Dominic D’Agostino and Pat Jak (who did the indirect calorimetry) in the summer of 2013. (I wrote this up immediately, but I’ve only got around to blogging about it now.) Dom is, far and away, the most knowledgeable person on the topic of exogenous ketones. Others have been at it longer, but none have the vast experiences with all possible modalities (i.e., esters versus salts, BHB versus AcAc) and the concurrent understanding of how nutritional ketosis works. If people call me keto-man (some do, as silly as it sounds), they should call Dom keto-king.
- I have tried the following preparations of exogenous ketones: BHB monoester, AcAc di-ester, BHB mineral salt (BHB combined with Na+, K+, and Ca2+). I have consumed these at different concentrations and in combination with different mixing agents, including MCT oil, pure caprylic acid (C8), branch-chained amino acids, and lemon juice (to lower the pH). I won’t go into the details of each, though, for the sake of time.
- The ketone esters are, hands-down, the worst tasting compounds I have ever put in my body. The world’s worst scotch tastes like spring water compared to these things. The first time I tried 50 mL of BHB monoester, I failed to mix it with anything (Dom warned me, but I was too eager to try them to actually read his instructions). Strategic error. It tasted as I imagine jet fuel would taste. I thought I was going to go blind. I didn’t stop gagging for 10 minutes. (I did this before an early morning bike ride, and I was gagging so loudly in the kitchen that I woke up my wife, who was still sleeping in our bedroom.) The taste of the AcAc di-ester is at least masked by the fact that Dom was able to put it into capsules. But they are still categorically horrible. The salts are definitely better, but despite experimenting with them for months, I was unable to consistently ingest them without experiencing GI side-effects; often I was fine, but enough times I was not, which left me concluding that I still needed to work out the kinks. From my discussions with others using the BHB salts, it seems I have a particularly sensitive GI system.
The hypothesis we sought out to test
A keto-adapted subject (who may already benefit from some Delta G arbitrage) will, under fixed work load, require less oxygen when ingesting exogenous ketones than when not.
Posed as a question: At a given rate of mechanical work, would the addition of exogenous ketones reduce a subject’s oxygen consumption?
The “experiment”
- A keto-adapted subject (me) completed two 20-minute test rides at approximately 60% of VO2 max on a load generator (CompuTrainer); such a device allows one to “fix” the work requirement by fixing the power demand to pedal the bike
- This fixed load was chosen to be 180 watts which resulted in approximately 3 L/min of VO2—minute ventilation of oxygen (this was an aerobic effort at a power output of approximately 60% of functional threshold power, FTP, which also corresponded to a minute ventilation of approximately 60% of VO2 max)
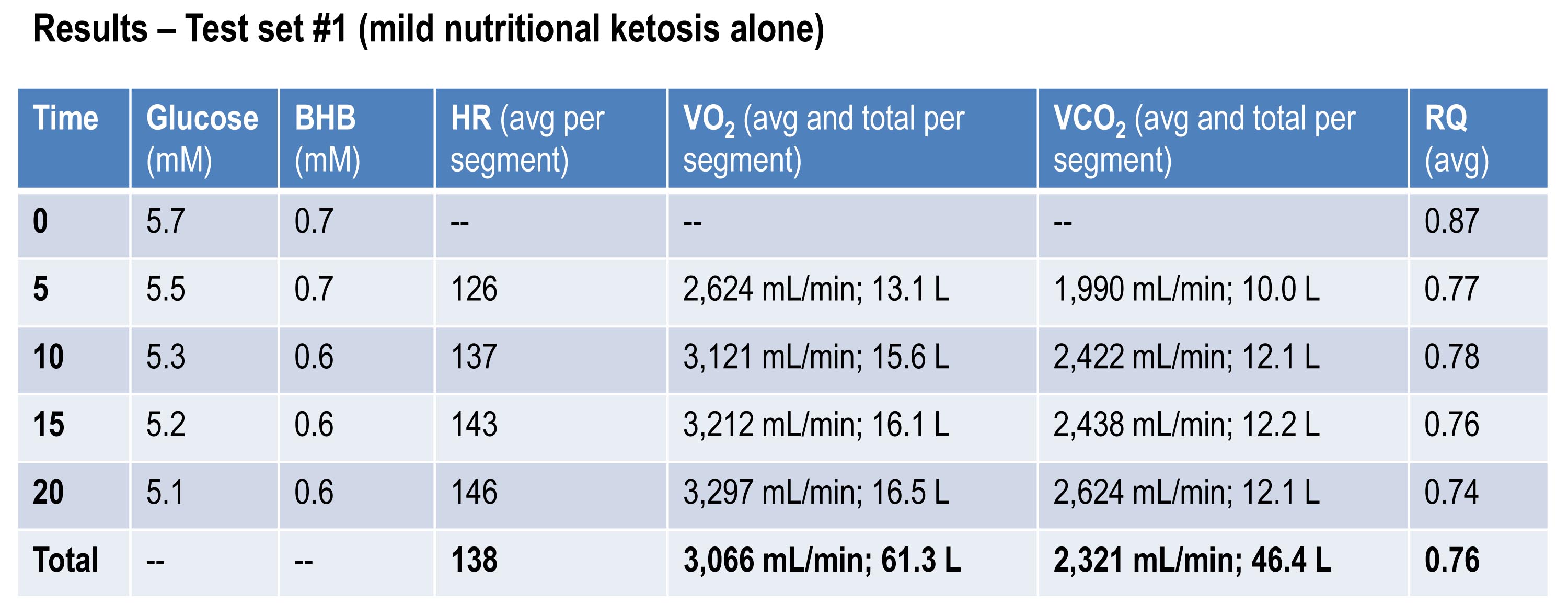
- Test set #1—done under conditions of mild nutritional ketosis, while still fasted
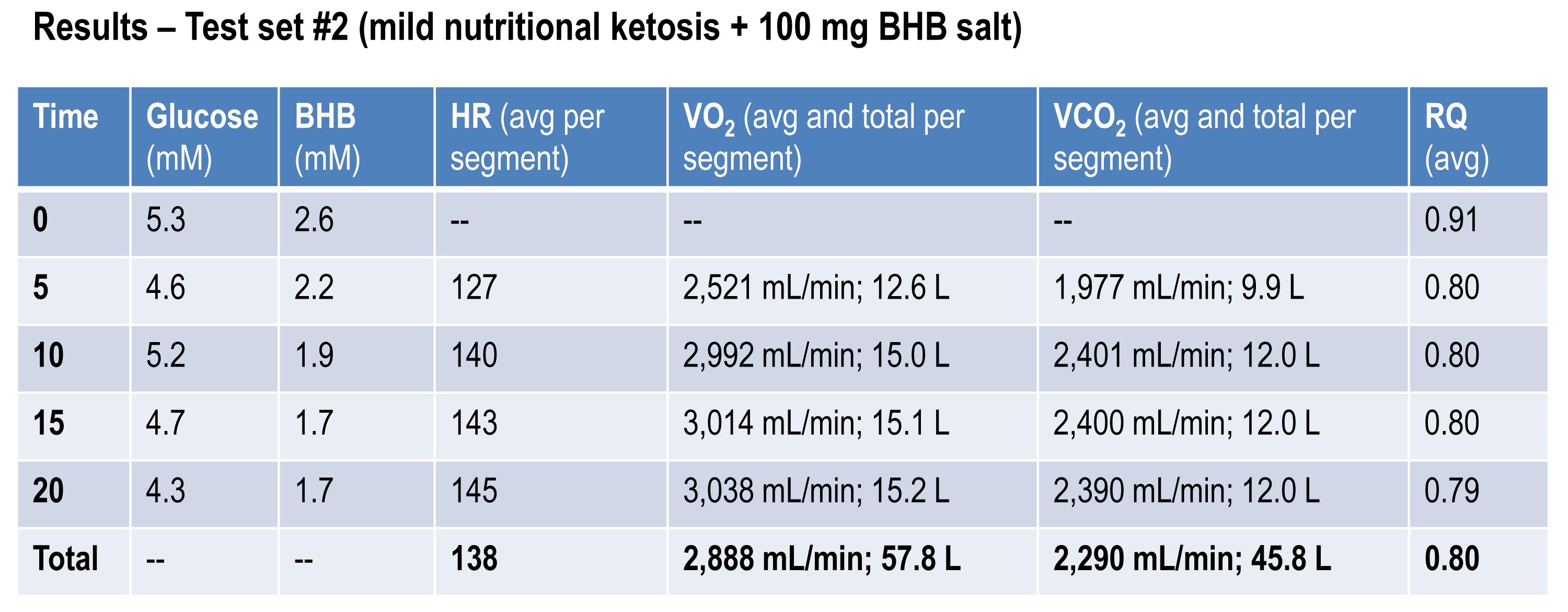
- Test set #2—60 minutes following ingestion of 15.6 g BHB mineral salt to produce instant “artificial ketosis,” which took place immediately following Test set #1
- Measurements taken included whole blood glucose and BHB (every 5 minutes); VO2 and VCO2 (every 15 seconds); HR (continuous); RQ is calculated as the ratio of VO2 and VCO2. In the video of this post I explain what VO2, VCO2, and RQ tell us about energy expenditure and substrate use—very quickly, RQ typically varies between about 0.7 and 1.0—the closer RQ is to 0.7, the more fat is being oxidized; the reverse is true as RQ approaches 1.0
Results
Test set #1 (control—mild nutritional ketosis)
The table below shows the data collected over the first 20 minute effort. The 20 minute effort was continuous, but for the purpose of presenting the data, I’ve shown the segmental values—end of segment for glucose and BHB; segment average for HR, minute ventilation (in mL per min), and RQ; and segment total for minute ventilation (in liters).
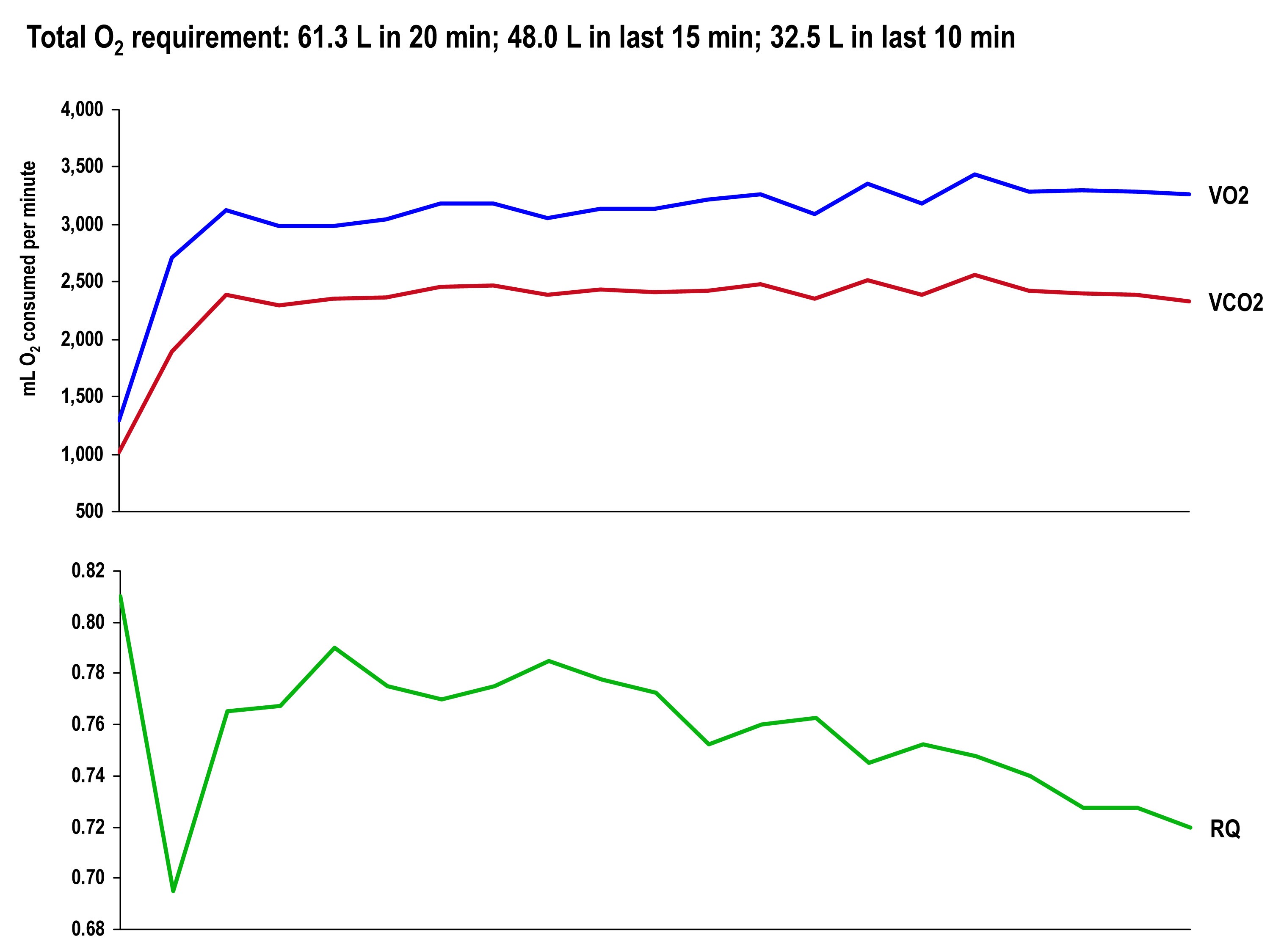
Glucose and BHB went down slightly throughout the effort and RQ fell, implying a high rate of fat oxidation. We can calculate fat oxidation from these data. Energy expenditure (EE), in kcal/min, can be derived from the VO2 and VCO2 data and the Weir equation. For this effort, EE was 14.66 kcal/min; RQ gives us a good representation of how much of the energy used during the exercise bout was derived from FFA vs. glucose—in this case about 87% FFA and 13% glucose. So fat oxidation was approximately 12.7 kcal/min or 1.41 g/min. It’s worth pointing out that “traditional” sports physiology preaches that fat oxidation peaks in a well-trained athlete at about 1 g/min. Clearly this is context limited (i.e., only true, if true at all, in athletes on high carb diets with high RQ). I’ve done several tests on myself to see how high I could push fat oxidation rate. So far my max is about 1.6 g/min. This suggests to me that very elite athletes (which I am not) who are highly fat adapted could approach 2 g/min of fat oxidation. Jeff Volek has done testing on elites and by personal communication he has recorded levels at 1.81 g/min. A very close friend of mine is contemplating a run at the 24 hour world record (cycling). I think it’s likely we’ll be able to get him to 2 g/min of fat oxidation on the correct diet.
The graph, below, shows the continuous data for VO2, VCO2 (measured), and RQ (calculated).
Test set #2 (ingestion of 15.6 g BHB salt 60 minutes prior)
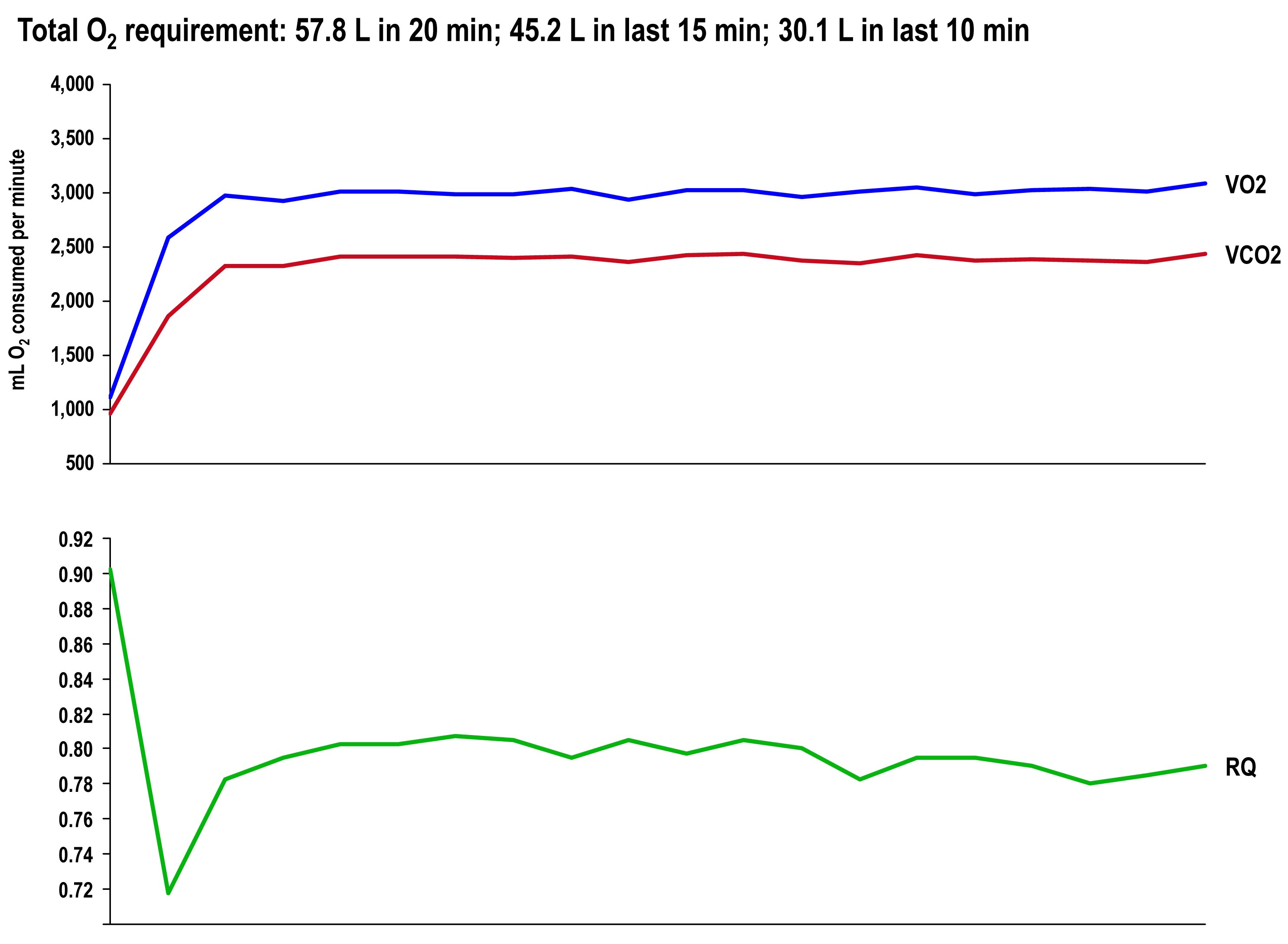
The table below shows the same measurements and calculations as the above table, but under the test conditions. You’ll note that BHB is higher at the start and falls more rapidly, as does glucose (for reasons I’ll explain below). HR data are almost identical to the control test, but VO2 and VCO2 are both lower. RQ, however, is slightly higher, implying that the reduction in oxygen consumption was greater than the reduction in carbon dioxide production.
If you do the same calculations as I did above for estimating fat oxidation, you’ll see that EE in this case was approximately 13.92 kcal/min, while fat oxidation was only 67% of this, or 9.28 kcal/min, or 1.03 g/min. So, for this second effort (the test set) my body did about 5% less mechanical work, while oxidizing about 25% less of my own fat. The majority of this difference, I assume, is from the utilization of the exogenous BHB, and not glucose (again, I will address below what I think is happening with glucose levels).
The graph once again shows the continuous data for VO2, VCO2 (measured), and RQ (calculated).
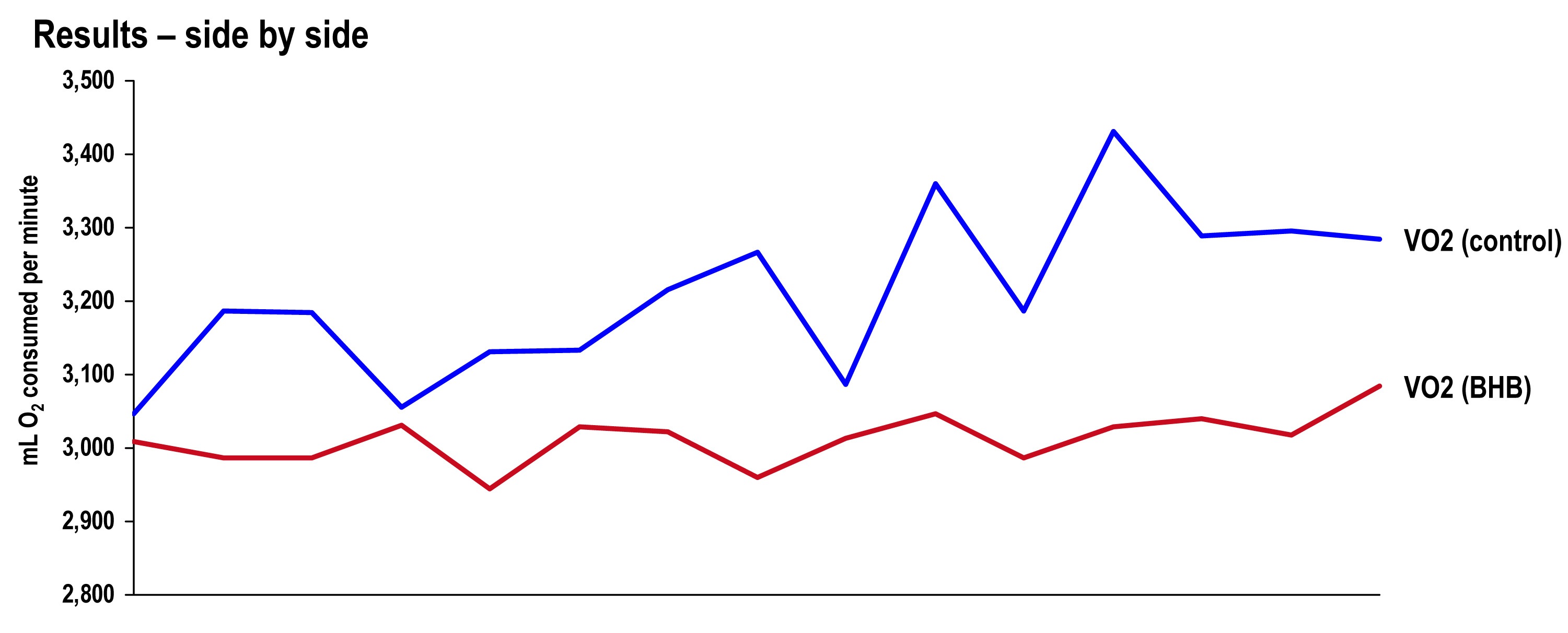
Side-by-side difference
The final graph, below, shows the continuous data for only VO2 side-by-side for the 20 minute period. The upper (blue) line represents oxygen consumption under control conditions, while the lower line (red) represents oxygen consumption following the BHB ingestion. In theory, given that the same load was being overcome, and the same amount of mechanical work was being done, these lines should be identical.
The hypothesis being tested in this “experiment” is that they would not be the same. Beyond visual inspection, the difference between the lines appears to grow as the test goes on, which is captured in the tabular data showing 5 minute segmental data.
Limitations
The most obvious limitation of this endeavor is the fact that it’s not an appropriately controlled experiment. Putting that aside, I want to focus on the nuanced limitations—which don’t impact the primary outcome of oxygen consumption—even if one were appropriately doing a real experiment.
- It’s not clear that the Weir coefficients used to estimate EE are relevant for someone in ketosis, let alone someone ingesting exogenous BHB. (The Weir formula states that EE is approximated by 3.94 * VO2 + 1.11 * VCO2, where VO2 and VCO2 are measured in L/min; 3.94 and 1.11 are the Weir coefficients, and they are derived by tabulating the stoichiometry of lipid synthesis and oxidation of fat and glucose and calculating the amount of oxygen consumed and carbon dioxide generated.) While this doesn’t impact the main observation—less oxygen was consumed with higher ketones—it does impact the estimation of EE and substrate use.
- In addition to the Weir coefficients being potentially off (which impacts EE), the RQ interpretation may be incorrect in the presence of endogenous or exogenous ketones. As a result, the estimation of fat and glucose oxidation may be off (though it’s directionally correct). That said, the current interpretation seems quite plausible—greater fat oxidation when I had to make my ketones; less when I got my ketones for “free.”
Observations from this “experiment” (and my experience, in general)
Animal models (e.g., using rat hearts) and unpublished case reports in elite athletes suggest supplemented BHB produces more ATP per unit carbon and per unit oxygen consumed than glycogen and FFA. This appears to have been the case in my anecdotal exercise.
The energy necessary to perform the mechanical work did not appear to change much between tests, though the amount of oxygen utilization and fat oxidation did go down measurably. The latter finding is not surprising since the body was not sitting on an abundant and available source of BHB—there was less need to make BHB “the old fashioned way.”
As seen in this exercise, glucose tends to fall quite precipitously following exogenous ketone ingestions. Without exception, every time I ingested these compounds (which I’ve probably done a total of 25 to 30 times), my glucose would fall, sometimes as low as 3 mM (just below 60 mg/dL). Despite this, I never felt symptomatic from hypoglycemia. Richard Veech (NIH) one of the pioneers of exogenous ketones, has suggested this phenomenon is the result of the ketones activating pyruvate dehydogenase (PDH), which enhances insulin-mediated glucose uptake. (At some point I will also write a post on Alzheimer’s disease, which almost always involves sluggish PDH activity —in animal models acute bolus of insulin transiently improves symptoms and administration of exogenous ketones does the same, even without glucose.)
In addition, the body regulates ketone production via ketonuria (peeing out excess ketones) and ketone-induced insulin release, which shuts off hepatic ketogenesis (the liver making more ketones when you have enough). The insulin from this process could be increasing glucose disposal which, when coupled with PDH activation, could drive glucose levels quite low.
If that explains the hypoglycemia, it would seem the absence of symptoms can be explained by the work of George Cahill (back in the day; see bottom figure in this post)—when ketone levels are high enough they can dominate brain fuel, even ahead of glucose.
Finally, these compounds seemed to have a profound impact on my appetite (they produced a strong tendency towards appetite suppression). I think there are at least two good explanations for this, which I plan to write about in a dedicated post. This particular topic—appetite regulation—is too interesting to warrant anything less.
Open questions to be tested in real experiments
- Are these results reproducible? If so, how variable are the results across individuals (by baseline metabolic state, diet, fitness)?
- Would the difference in oxygen consumption be larger (or smaller) in an athlete not already keto-adapted (i.e., not producing endogenous ketones)?
- Would the observed effect be greater at higher plasma levels of BHB (e.g., 5 to 7 mM), which is “easily” achievable with exogenous ketones?
- Would the observed effect be the same or different at higher levels of ATP demand (e.g., at FTP or at 85-95% of VO2 max)?
- Would the trend towards improved energy efficiency continue if the exercise bout was longer in duration (say, greater than 2 hours)?
- How will exogenous ketones impact exercise duration and lactate buffering?
- Why do exogenous ketones (both BHB and AcAc it seems) reduce blood glucose levels so much, and can this feature be exploited to treat type 2 diabetes?
- Are there deleterious effects from using exogenous ketones, besides GI side-effects?
- What are the differences between exogenous BHB and AcAc (which in vivo exist in a reversible equilibrium) on this particular phenomenon? (Work by Dom D’Agostino’s group and others have shown other differences in metabolic response and clinical application, including their relative impact on neurons.)
Photo by Alexey Lin on Unsplash







I have a quick question about exogenous ketones. When you take exogenous ketones and eat carbs what does your body do with the carbohydrates in your system? Does it cause elevated blood glucose because your body is not using that as your source of energy?
look for delauer video on that.. shortly said not good…you might store the glucose while using the ketones.
Can you do high intensity interval training (HIIT) and get good results in a ketogenic state? I’ve been low carb for about a year and ketogenic for the past 5-6 months.
When a person takes exogenous ketones, do the body stop or reduce the amount of ketones it produces? And, if a person regularly supplements with exogenous ketones, would the body take time to resume normal production levels, similar to the way brain chemical production is reduced in people who abuse drugs? Thanks!
I take care of several athlete “extremists” who, despite having very lean bodies, have elevation of A1C and even fasting glucose. Insulin is often in the 5 to 10 range…….higher than I’d expect for an elite athlete with extreme insulin sensitivity. My question is: Can “over” exercising lead to a degree of insulin resistance. In the ketotic state….or exogenously provided ketones……I see the glucose fall during exercise…..but in the carb loaders who aren’t fat adapted……I’m guessing glucose production during exercise (some of these guys ride at an average wattage of 230-250 for hours) exceeds uptake?
Hi Peter,
After seeing my MD lose 60 lbs on the IdealProtein diet in 4 months (LCLFModP Ketogenic), I started the program, and have lost 13 lbs in 2 weeks. Easy and enjoyable. I use a Ketonix Sport daily and typically show 7-Green with a few days of 5-Yellow mixed in. I used KetoCaNa (Amazon) one morning before heading to the gym and the Ketonix flashed Red showing high traces of ketones. I felt an increase in energy and clarity throughout the day even on just a half serving. I can attest to the efficacy of using exogenous ketones for added energy, and I wonder if it will help me get fat-adapted quicker. Unsure if I will use KetoCaNa regularly, or just before workouts, or not at all until I hit my target weight.
Thanks for all your research and writing. I really appreciate learning from you in this exciting field.
Hey Peter,
I was wondering if you had any thoughts about the most recent NuSI study that was headed by Kevin Hall. Thanks.
Hi Peter,
Thanks for using yourself as a guinea pig and thereby creating radials for future explorations.
My friend introduced exogenous ketones to me and I just started googling and yours is the first article that came up. Suuuuper interesting! I love exercising but you’d never know it to look at me. I wish my body would burn the calories rather than storing them and leaving me tired and hungry. I’m less concerned about my appearances than I am about not having energy and the increasing load the extra weight puts on my system. However I do notice that, not only for myself, I never met a weight loss regime that didn’t have the tendency messing with my energy levels and with the end result of developing a more efficient metabolic system i.e. being larger than before a couple years later.
If you have any recommendations for websites that are aimed at newbies like me, I’d appreciate the link.
I like your writing clarity and will check out the rest of your site.
Brenda
My daughter did the ketosis diet and had remarkable results. As with anything that works, the supplement and MLM folks were soon to follow. So I have friends pushing various ingestible “ketones” products. Are those similar to what you were taking, and can breaking down food into micro-nutrients really work?
I had Chronic Obstructive Pulmonary Disease (COPD) for 7 years, My first symptoms were dry cough, chest tightness and shortness of breath. This was ongoing for 3 years, my first chest x-ray only showed bronchitis. Finally I went to a pulmonologist and was diagnosed with COPD, I was immediately placed on Seebri Breezhaler, it’s an inhalation powder, I take it once a day in the morning. Finally, i started on COPD Herbal Formula i ordered from NewLife Herbal Clinic, the herbal formula worked 10x better than all the medications my pulmonologist had prescribed. I have had a total decline of symptoms since i started on this herbal formula, visit www .newlifeherbalclinic .com or email info@ newlifeherbalclinic. com
Larry HEMM
I had Chronic Obstructive Pulmonary Disease (COPD) for 7 years, My first symptoms were dry cough, chest tightness and shortness of breath. This was ongoing for 3 years, my first chest x-ray only showed bronchitis. Finally I went to a pulmonologist and was diagnosed with COPD, I was immediately placed on Seebri Breezhaler, it’s an inhalation powder, I take it once a day in the morning. Finally, i started on COPD Herbal Formula i ordered from NewLife Herbal Clinic, the herbal formula worked 10x better than all the medications my pulmonologist had prescribed. I have had a total decline of symptoms since i started on this herbal formula, visit www .newlifeherbalclinic .com or email info@ newlifeherbalclinic. com
Larry HEMM
I’ve been on a keto diet for quite some time for weigh loss purposes.
I was looking in to possible weigh loss benefits of exogenous ketone supplements.
I hadn’t really delved too far in to the science of this and your article and self-experiment are quite fascinating and appear to be far more controlled than typical home-brew experiments. Further study under more rigorous conditions is warranted.
One interesting use of this could be as it applies to space science/travel: Oxygen is expensive to lift in to orbit and not terribly efficient to recycle. The less oxygen used by space travelers, the better. Could a ketone-rich diet help astronauts use less oxygen?
Peter et al….
Has anyone experienced or heard of anyone having heart problems leading to seizure, collapse, heart attack, and/or cardiac arrest they could trace to an overdose of Exogenous Ketones?
I believe I overdosed on KetoCana (perhaps as much double the standard dose) as I was finishing up the remainder of my supply I had bought from a friend who had previously purchased the KetoCana. This would not likely have been a simple averse reaction as I had been experimenting/using Exogenous Ketones (including KetoCana, Keto/OS, etc) for a couple months already (less than 10 times though total).
My story is that on May 20th I may have double dosed on the KetoCana, when I took it in between a double work shift I was doing that day/night. My food intake was low and I was doing an intermittent fast but that was a regular thing for me. However, I apparently came through the 2nd shift with more energy than normal as a girlfriend I was with later that night commented about. Also I acted sassier than normal, not sure if that’s a symptom or I simply had extra energy and chose to use it that way.
The next morning (after very little sleep) I woke up to attend a martial arts class and behaved strangely when I arrived, not talking to or greeting other students or the master/shifu (not normal behavior for me) and I began a slow jogging warmup. Apparently after less than a single lap around a small gym, I headed towards the other room and bumped into a plant and collapsed (possibly having a seizure), my heart got up to 200 beats/minute and then stopped. I was given CPR for 10 minutes, ambulance arrived and shocked me twice, I was cooled and put in coma at hospital and came out of it a few days later amazingly with no apparent brain damage (except acute memory loss extending from the day I took the Ketones (day before collapse) to the day after I woke from the coma. Apart from that, it’s been over 3 weeks since my release from cardiac care unit and my memory is as sharp as ever (minus the incident and the timeframe mentioned before which is completely blank still). My other cognitive abilities seem normal as well. On one hand I think all my Keto-type eating, C8, Coconut Oil, and Exogenous Ketones may be responsible for protecting my brain and on the other hand I can’t think of any other reason the heart attack could have been triggered unless it was simply long term lack of sleep but that’s nothing new for me. I’m 40 years old and have been an active athlete all my life and this is the first heart-related incident I can recall ever having. Anybody think it could have been overdosing on Exogenous Ketones (KetoCana)?
PS>..I was also taking lots of other supplements like Rhodiola, Maca, Matcha green tea but from my containers from that day it didn’t look like I’d had time to consume much of anything yet that morning and no I didn’t consume any ketones that morning either. Just the afternoon before. Could they even still have been in my system to cause a heart problem??
Anyone that has any thoughts they are much appreciated as I work to figure out what happened and try to determine if it’s safe for me to bring back ketones into my diet after this incident. Thanks again!
-Darren
All,
I have had the same low glucose after endurance exercise. After a 70 mile group ride here in hot Houston summer, drinking water with ketone salts, my glucose was 67 mg/dL and Ketones were 2.2 mmol/L with plenty of energy and no mental fog or nausea. I do not recall the quantity of BHB consumed but it was minimal (say at half recommended). The fun part is that I punched out a killer sprint off the front (of a modest 20 mph group) and I was holding 30 mph all alone (I’m complaining, not bragging…) for 10-15 seconds until I thought “this has to be bad for me” and slowed down. So perhaps this was the 5% improvement in O2 utilization suggested (felt like more than that). It might also be the awesome job of cleaning and lubing I did on my chain the night before. So usually a “redline” like that messes up the rest of the ride. Not in this case. The unflavored ketone water tastes pretty good when you are thirsty, not so much sipping it all day long.
FYI this is a paper linking exogenous BHB as a possible “longevity in a pill” candidate (makes me think of the glucose suppression aspect):
https://www.ncbi.nlm.nih.gov/pubmed/28371201
Cheers and keep your eyes wide open on the road.
question 1: are you saying that using exogenous BHB’s versus endogenously induced ketosis will not promote body fat burning? (from a long time low carber who would like to try cheating…)
Question 2: son has CTE from a severe TBI & symptoms improve dramatically on MCT’s & ex BHB’s, Any research on this application? only see stuff for AD
I’ve got a business Ph.D. and I used to be a certified fitness instructor. Most every semester my ethics class does the Lance Armstrong Harvard Case Study. The athletes and the exercise sports science people puzzle over Lance’s supposedly greater V02 Max reported as part of the case. After seeing this ‘experiment’ I wonder if Lance didn’t have better ketone availability? Should that even be a question? It’s a rhetorical question in the end, the real question lies in whether or not Lance believed he was cheating…clearly no.
I likewise conceive hence, perfectly indited post!
https://osf.io/preprints/nutrixiv/rdjfb/?fbclid=IwAR1sZW6xGVAuAJi5bJs9DG7kAE0oHn-92huFnQZyqzKY_BKZOzPgcVLxF70
Please react or respond to this.
Thank you.
Hi Dr. Attia!
I’ve been in ketosis for over a year and believe I’m fat adapted because I don’t crave sugars or carbs and can go long periods of time without eating and still not be hungry. However, while my energy isn’t bad, it isn’t as good as it is compared to when I am on carbohydrates. Is this normal, or should I try going deeper in ketosis? Right now my ketone levels consistently measure “medium” to “light” on ketone strips, is it possible that I should get to even deeper levels of ketosis and stay there in order to improve energy levels further? If it helps, I would describe my life to be very low activity (exercise, sports etc) I’m wondering what your opinion is considering all of the experience you have with this. Thanks for all your information and knowledge!
Thanks Peter, I’ve gotten a real sick kick out hearing the original story on Tim Ferriss and others podcasts about the ‘jet fuel’ experience.
Now that’s dedication!