Benoît Arsenault is a research scientist focused on understanding how lifestyle and genetic factors contribute to cardiovascular disease risk. In this episode, the discussion casts a spotlight on Lp(a)—the single most important genetically-inherited trait when it comes to atherosclerotic cardiovascular disease (ASCVD) risk. Benoît explains the biology of Lp(a), how it’s inherited, the importance of measuring Lp(a) levels, and the diseases most associated with high Lp(a). He dives into data on the possible treatments for lowering Lp(a) such niacin, statins, and PCSK9 inhibitors, as well as the most exciting new potential therapeutic—antisense oligonucleotides.
Subscribe on: APPLE PODCASTS | RSS | GOOGLE | OVERCAST | STITCHER

We discuss:
- How Benoît came to study Lp(a)—a new marker for cardiovascular risk [3:15];
- The relationship between Lp(a) and CVD risk [6:45];
- What genome-wide association studies (GWAS) revealed about Lp(a) [16:00];
- Clinical tests to measure Lp(a) [22:00];
- The biology of Lp(a) [25:45];
- How statins lower LDL-cholesterol and why this doesn’t work for an Lp(a) [29:15];
- The structure of LDL-p and Lp(a) and what makes Lp(a) more atherogenic than an equivalent LDL particle [34:00];
- The role of Lp(a) in aortic valve disease [42:45];
- How greater numbers of Lp(a) particles are associated with increased risk of disease [48:00];
- The genetics and inheritance of Lp(a) and how and when to measure Lp(a) levels [52:00];
- Niacin and other proposed therapies to lower Lp(a), apoB, and CVD risk [1:00:45];
- Why awareness of Lp(a) among physicians remains low despite the importance of managing risk factors for ASCVD [1:14:00];
- The variability of disease in patients with high Lp(a) [1:19:00];
- Diseases most associated with high Lp(a) [1:26:30];
- The biology of PCSK9 protein, familial hypercholesterolemia, and the case for inhibiting PCSK9 [1:35:00];
- The variability in PCSK9 inhibitors’ ability to lower Lp(a) and why we need more research on individuals with high levels of Lp(a) [1:50:30];
- Peter’s approach to managing patients with high Lp(a), and Benoît’s personal approach to managing his risk [1:54:45];
- Antisense oligonucleotides—a potential new therapeutic for Lp(a) [1:57:15]; and
- More.
Get Peter’s expertise in your inbox 100% free.
Sign up to receive An Introductory Guide to Longevity by Peter Attia, weekly longevity-focused articles, and new podcast announcements.
How Benoît came to study Lp(a)—a new marker for cardiovascular risk [3:15]
Benoît’s training
- Benoît got involved in Lp(a) research during his postdoc years
- He trained at Laval University in Quebec City from 2006-2009
- Did work in the field of lipids, looking at LDL particle size, triglycerides, APOB, etc., on cardiovascular outcomes
- Not that many people talked about Lp(a) because there had been so many negative studies on Lp(a) and the risk of cardiovascular disease
- Such as myocardial infarction (MIs) and stroke and so on
Studies of biomarkers associated with cardiovascular events point to Lp(a)
- Genetic association studies have resurrected the field of Lp(a)
- These were published in 2009 to 2011
- During that time he was a postdoc in Amsterdam working with John Kastelein
- John was working on the treating-to-new-targets trial, the TnT trial
- This is one of the first trials that showed that if you reduce low density lipoprotein (LDL) levels by increasing the statin dose, you’ll get an incremental benefit in cardiovascular outcomes
- They were working on a bunch of sub analyses for that trial
- They had just measured a whole panel of emerging biomarkers that could be associated with cardiovascular events like CRP, NT-proBNP, inflammatory markers, other biomarkers of insulin sensitivity, and Lp(a)
- And it turned out that of a huge list of 18 biomarkers that they had measured in thousands of individuals, Lp(a) was actually the strongest of them that was predicting residual cardiovascular risk
- This was the first paper he published on Lp(a) in those years
- Published in the Journal of the American College of Cardiology, Oxidation-Specific Biomarkers, Lipoprotein(a), and Risk of Fatal and Nonfatal Coronary Events
- At that time they were also wondering if there were any genetic variants that could explain statin response
- Because statins work well in most people, but there’s a huge inter-individual variability in terms of LDL lowering associated with statins
- They were first used to identify genetic variants associated with specific diseases
- They were part of a big genetic consortium that was called the GIST consortium (genomic investigation of statin therapy)
- It turned out from that big analysis that Lp(a) was the most important genetic risk factor that explains statin response
- They showed that if you have high Lp(a), then your LDL wouldn’t be lowered as much as if you didn’t have a high Lp(a)
The relationship between Lp(a) and CVD risk [6:45];
Epidemiological studies show the importance of Lp(a)
- Ballpark numbers are that about 20% of the world population has an Lp(a) level that puts them in a higher risk category
- Individuals of African ancestry have the highest levels while Chinese and Japanese probably have the lowest levels
- Lp(a) was discovered in 1963 by a Swedish scientist named Kare Berg
- Kare Berg was one of the first to show in the late 70s – early 80s that Lp(a) was associated with cardiovascular events, in European cohorts
- The assays to measure Lp(a) were not as good then as they are today; there was a lot of variability
- A lot of these studies published in the 90s and in the early 2000s came out negative, so nobody talked about Lp(a)
Why were initial studies of Lp(a) negative?
- The hypothesis was that high Lp(a) was associated with cardiovascular events like myocardial infarction, stroke, etc.
- People with higher Lp(a) levels were at the highest risk
- This turned out to NOT be the case in many of those studies
- They realized afterwards that the assays used were probably not very good
- The assays did not correctly identify people with high Lp(a)
- This has been attributed to the complex structure of lipoprotein(a), copy number variation in the Lp(a) genes
- And the antibodies that are used against that, sometimes they can bind to different epitopes of Lp(a)
- So now we have antibodies that are binding to Lp(a) on other regions
- This provides a much better sense of the number of Lp(a) particles in the bloodstream
- But back in the days, the assays were overestimating the isoform size that was bigger, and they were underestimating the small Lp(a) isoform size
- It’s the small Lp(a) isoform size which is associated with high Lp(a)
- The Lp(a) gene was cloned in the 80s by Angelo Scanu’s group at the University of Chicago
- A PubMed search of Lp(a) shows a straight line form the 70s-mid 80s
- Then when these studies came back negative, the number of studies listed on PubMed goes down
- They come back up in 2009/ 2010 when genetic association studies were beginning to be published
- The great thing about genetic studies is that you don’t necessarily need to measure lipoprotein(a) levels
- Now the number of studies on Lp(a) listed on PubMed are going back up again
- There were 3 big studies published in 2009 that very convincingly shows that genetic variants associated with high Lp(a) levels were tracking with cardiovascular events
- Phenotypic studies of individuals with lower Lp(a) levels (1 standard deviation below the mean) show reduced risk for 5 cardiometabolic diseases, shown in the figure below
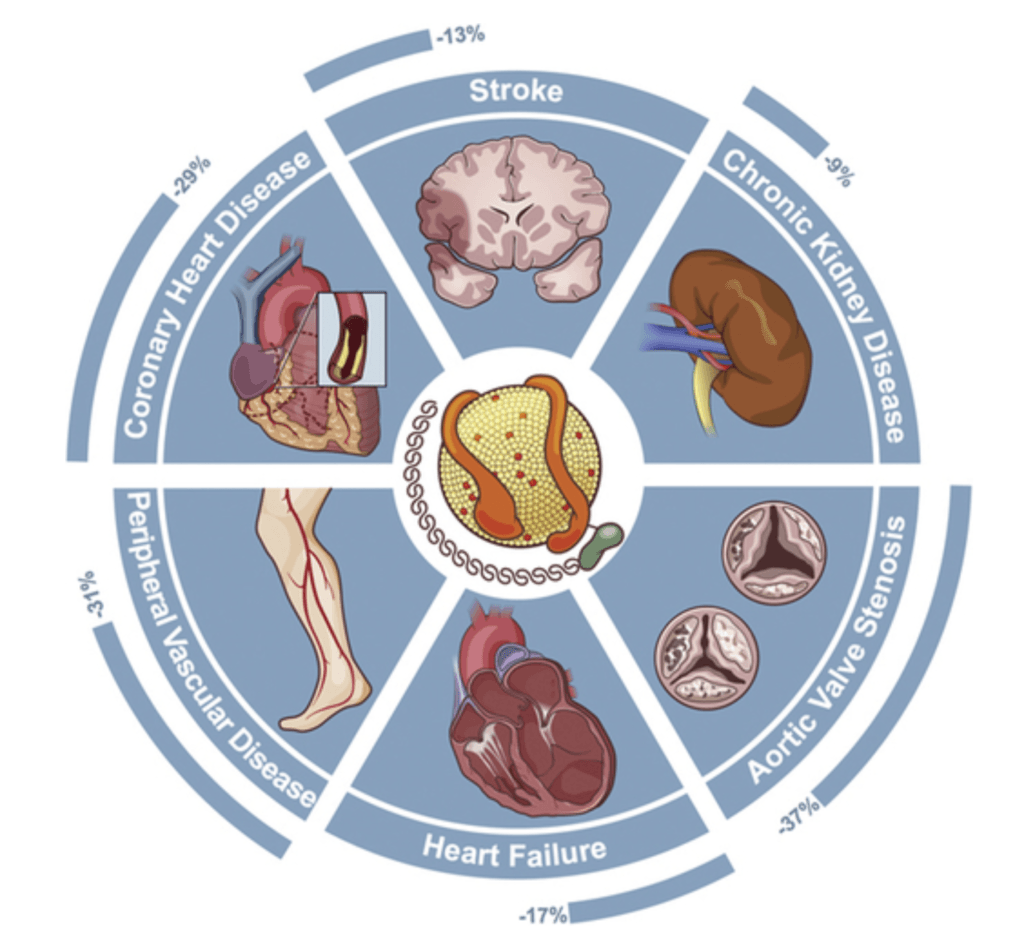
Figure 1. Lower Lp(a) levels associated with reduced risk of 5 cardiometabolic diseases. Image Credit: Journal of the American College of Cardiology 2016
{end of show notes preview}
Would you like access to extensive show notes and references for this podcast (and more)?
Check out this post to see an example of what the substantial show notes look like. Become a member today to get access.

Benoît Arsenault, Ph.D.
Benoît Arsenault obtained his doctoral degree in Physiology-Endocrinology from Université Laval in Québec City, Canada in 2009. He did postdoctoral research at the Academic Medical Center in Amsterdam and at the Montreal Heart Institute. Benoît is currently an Assistant Professor in the Department of Medicine at the Université Laval and a research scientist in the cardiology axis at the Centre de recherche de l’Institut universitaire de cardiologie et de pneumologie de Québec, in Canada.
The research of Benoît’s team is focused on understanding the risk of cardiovascular diseases such as atherosclerosis and aortic stenosis in relation to lifestyle and inherited risk factors. This includes extensive research in unraveling the role of Lp(a), HDL metabolism, PCSK9, and lipid-lowering therapies. [iucqp.qc.ca]