Hussein Yassine is a physician and researcher who studies brain lipid utilization in the context of finding preventative measures for cognitive impairment, specifically Alzheimer’s disease (AD). In my conversation with Hussein, we begin with a fundamental coursework in brain biology—including its architecture and energy systems. We go on to discuss what these systems look like when something goes wrong and cognitive decline ensues. We talk about the evolutionary origins of the ApoE genotype, with specific attention to the ApoE4 allele and its association with AD. We spend time discussing ApoE4 implications for the brain’s fuel utilization, notably omega-3 fatty acids: EPA and DHA. We briefly pivot to the implications of recent omega-3 trials for cardiovascular disease and return to what we currently understand about EPA/DHA and brain health; we contemplate potential dietary interventions across the lifespan to preserve and prolong cognitive function.
Subscribe on: APPLE PODCASTS | RSS | GOOGLE | OVERCAST | STITCHER
We discuss:
- Hussein’s Background and introduction to brain composition (3:00);
- The blood-brain barrier and brain filtration (8:00);
- Lipids and brain function (13:00);
- How the brain utilizes energy (18:00);
- Apolipoprotein E (ApoE) structure and function in the periphery (27:30);
- ApoE function in the brain (38:15);
- Evolutionary origins of ApoE isoforms (43:45);
- ApoE4 variant and Alzheimer’s Disease (AD) risk (53:30);
- Dietary fuel preference with the ApoE4 allele (1:03:00);
- The role of omega-3 fatty acids in the brain (1:13:30);
- Comparing findings from the REDUCE-IT and STRENGTH trial (1:21:45)
- The relationship between dietary omega-3 intake and brain health (1:34:15);
- Preventing cognitive decline: A critical window for DHA in ApoE4 carriers? (1:42:30);
- Hussein’s ongoing research and recommendations for E4 carriers (1:54:00); and
- More.
Get Peter’s expertise in your inbox 100% free.
Sign up to receive An Introductory Guide to Longevity by Peter Attia, weekly longevity-focused articles, and new podcast announcements.Hussein’s Background and introduction to brain composition [3:00]
- Hussein grew up in Beirut, Lebanon and went to medical school there and moved to the U.S. to do residency training and his fellowship
- Did a fellowship in endocrinology
- Comes from a family with heart disease and diabetes so decided to subspecialize in lipid disorders
- He studied blood lipids for 2-3 before moving to brain lipids
The brain is composed of 3 main types of cells
- The brain is a unique compartment that is mostly lipid
- Contains a large amount of lipids—from sterols to different forms of fatty acids
- Neurons which are responsible for firing and forming synapses; the regulate how the brain functions
- Helper cells help neurons function in the brain
- glial cells are the immune cells responsible for clean up
- Microglial cells are cleanup specialists and have been linked to neurodegenerative diseases
- astrocytes repair and supply nutrients; regulate energy storage production
The blood-brain barrier and brain filtration [8:00]
The brain is surrounded by the blood-brain barrier
- separates the blood vessels and the blood inside the blood vessels from the brain
- designed to protect the brain and to maintain a stable environment
- composed of:
- endothelial cells called capillary cells that have a lining of tight junctions
- Pericytes surround those endothelial cells and support the maintenance of the barrier
- mural cells, or wall cells, make up the matrix surrounding pericytes and capillary cells
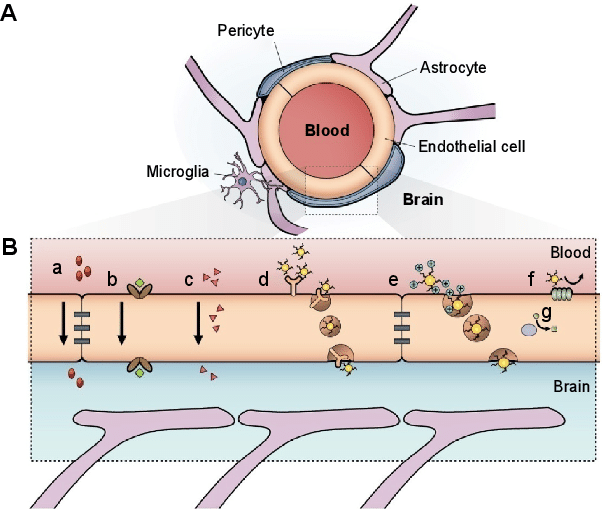
Figure 1. Schematic representation of the blood–brain barrier. (A) Scheme of the neurovascular unit that constitutes the BBB and (B) the main routes of the crossing of molecules to the CNS are represented [source]
Cerebral spinal fluid (CSF) os the “sewage system” of the brain
- Allows drainage from the brain cells, from the interstitial fluid
{end of show notes preview}
Would you like access to extensive show notes and references for this podcast (and more)?
Check out this post to see an example of what the substantial show notes look like. Become a member today to get access.

Hussein Yassine, M.D.
Hussein is a physician and researcher who is an Associate in the Department of Medicine, Keck School of Medicine of USC. His lab studies how changes in lipid metabolism and nutrition affect cognition and the risk of developing Alzheimer’s disease, paying specific attention to how the APOE4 allele affects lipid metabolism and response to diet. He directs the Roybal Alzheimer’s disease Research program focused on understanding how obesity, diabetes, and vascular risk factors in Los Angeles’s Latinx population affect cognition and Alzheimer’s disease risk. He also runs the Lipid Clinic at Roybal Healthcare Center in East LA, providing care for a large population of complex lipid and diabetes disorders that are referred from all LA county clinics.