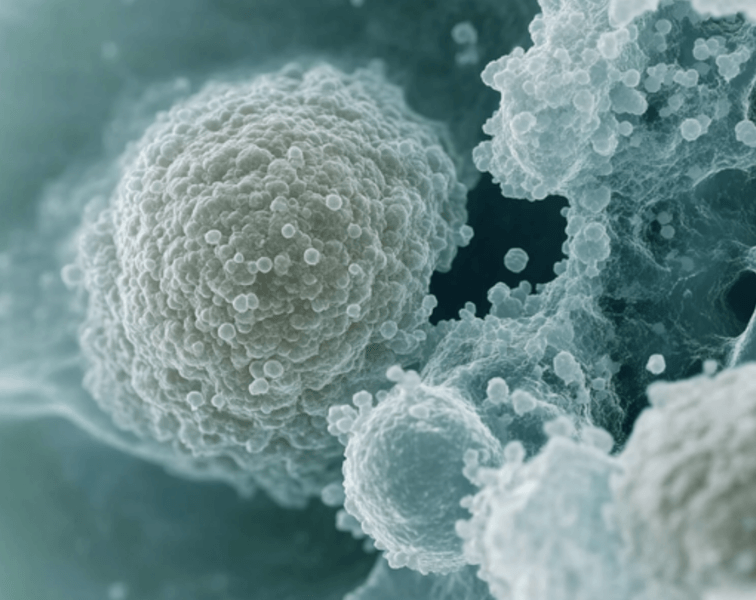
In this episode, Keith Flaherty, director of clinical research and targeted cancer therapy at Massachusetts General Hospital, shares his vast wealth of knowledge in cancer starting with the history of treatment from chemotherapy to radiation to surgical therapy and where those methodologies seemed to have leveled off. He also walks us through the timeline of advancements (and lack there of) from when the War on Cancer was declared in the 1970s, through the sequencing of the entire human genome, and all the way to today. Keith dives into the topic of immunotherapy, probably the most exciting recent development in cancer therapy, and also provides us a rundown of his notion of a different approach to cancer that attacks all the essential pillars of cancer growth and survival. Finally, we talk a little bit about liquid biopsies, we discuss the roles of CRISPR and other potentially over-hyped therapies with respect to cancer. We also touch on stem cell therapy a bit, as well as some other common cancer-related questions such as the role of vitamin D and sun exposure in melanoma, and much more.
Subscribe on: APPLE PODCASTS | RSS | GOOGLE | OVERCAST | STITCHER
We discuss:
- Growing up around medicine, and finding a career that you love [7:30];
- Medicine as a career, limitations of the med school teaching approach, and the dynamic and accelerating field of medicine and technology [16:30];
- Explaining chemotherapy, radiation, and how a cancer develops [23:45];
- Surgical oncology, cure rate of solid tumors, and survival rate after tumor removal [33:15];
- 25 years after the War on Cancer is declared, gene sequencing, and why Keith’s was fascinated by the HIV case study [37:15];
- Cancer immunotherapy: History, how it works, and why some cancers respond and others don’t [46:00];
- MHC complexes, and cancer cloaking mechanisms [56:00];
- Comparative biology of cancer: Why some cancer can evade immune detection better than others [1:03:00];
- What we learned from the Cancer Genome Atlas Project [1:07:00];
- Defining targeted therapy, HER2 breast cancer, chronic leukemia, and the translocation of chromosomes [1:12:00];
- Tumor protein P53, the most famous tumor suppressor gene and its ubiquity in cancer [1:17:45];
- Activated oncogenes, the RAS pathway, PI3 kinase, RAF gene, and Keith’s “aha moment” [1:24:15];
- Advice for starting your career as a scientist/clinician [1:37:00];
- Fusion-driven cancers, targeted therapy, and the Bcr-Abl/chronic myelogenous leukemia case study [1:39:45];
- Targeted therapy for fusion-driven solid tumors, adjuvant systemic therapy, and the HER2 breast cancer example [1:53:00];
- Advancing melanoma treatment, survival, and cure rates with BRAF-MEK combo therapy [1:59:15];
- The fundamental pillars of cancer growth and survival, and the toolkit we need to attack cancer from all angles [2:02:40];
- Peter’s clinical framework for thinking about cancer and how Keith might improve it, and how the biotech environment is hampering our ability to put together novel cancer treatments [2:05:00];
- How useful is CRISPR in terms of tumor suppressing? [2:16:15];
- Liquid biopsies as a therapeutic monitoring tool [2:18:00];
- Stem cell therapy: The efficacy and potential risks [2:25:15];
- Aging and cancer: Is cancer inevitable? [2:28:45];
- Vitamin D supplements, sun exposure, melanoma, and exercise [2:32:30];
- How and why Keith has straddled the line between science/research and industry/drug companies, and the importance of getting more voices of practitioners at the table [2:42:00]; and
- More.
Get Peter’s expertise in your inbox 100% free.
Sign up to receive An Introductory Guide to Longevity by Peter Attia, weekly longevity-focused articles, and new podcast announcements.Growing up around medicine, and finding a career that you love [7:30]
Where are you from?
- Born and raised in Baltimore
- Broke away to boarding school in Massachusetts for four years
- Connecticut for college, four years,
- then back to Baltimore, four years of medical school.
- Boston for the first time, three years of residency and
- then, Philadelphia for nine years, which was both medical oncology fellowship and my first faculty stint.
- And now, ten years ago, moved back up to Boston to Mass General.
Growing up around medicine
- Father and mother were physicians
- My father was an academic cardiologist for 25 years
- Mom was an academic psychiatrist, actually, really still is at the age of now
When did you know you wanted to go into medicine?
- Knew he wanted to “help” people is some way or another
- Didn’t want to go into the “business” world
- He also saw that both his parents were very passionate about what they were doing
- Eventually, medicine just seemed to stand out among the career options
The fine line for parents: “being home” vs. “setting an example” in terms of seeing them working on something with passion and discipline
- There’s something to be said when that child sees their mom or dad feeling incredibly passionate about what they’re doing
- For example, Keith’s wife (Mira Kautzky)
- She’s an internist at Mass General
- They watch her pour herself into the indirect care of patients through the electronic medical record and constantly reflect that she’s the hardest working person they know.
- That kind of willingness to give, give, give outside of one’s personal time, family time, is related to this very nebulous notion of what it is that qualified as a satisfying career
Keith feels luck to do what he does for a living
- Keith feels like most people dislike their job and they’re just holding their breath trying to get to the weekend
- He says he is part of that small group of people who love what they do
- That said, his work/life balance has been “off the rails” in terms of working long hours and being over committed and trying to juggle way too many things
- However, he would also say that “it was exactly the optimal amount of chaos that one needs to really feel like you’re pushing on all fronts simultaneously”
- How do you both find the outlet and define the scope of a career that allows you to do that? ⇒ “What a challenge!”
- Keith believes the changing times will likely create an even bigger challenge for future generations
Medicine as a career, limitations of the med school teaching approach, and the dynamic and accelerating field of medicine and technology [16:30]
Peter says, the path to “academic success”, non-academic clinical success, it’s still relatively unperturbed compared to so many other paths outside of medicine… does Keith agree?
- “I don’t see it that way” says Keith
- Keith sees how rapidly the technology and wealth of knowledge is accelerating
- Things like data generation, ability to produce high dimensional data, analyze it, try to make sense of it, raise hypotheses, test hypotheses, yet the cycle time is changing a lot, and the constituents that we now need to interact with are totally different
- The point I’m coming to… I see medicine now as this terrifying arena in which to try to assemble a multi-decade long career.
How do you keep yourself relevant in a 40 year arc? Keith posits…
- Here I am at 20 years, I feel like I’m an absolute dinosaur
- Keith wonders how the new generation of physicians and scientists will keep up with this rapidly accelerating progress
Peter’s take:
- Based on the way you’re describing it, Keith, I would say doctors coming out of training are hosed, because … I think medical school might be one of the most anti-intellectual forms of higher education that exists.
- Peter says even in undergraduate, law school, Ph.D. programs… you are encouraged to think creatively, and problem solve
- In other words, “I’m not sure medical school teaches you how to be a thinker or a problem-solver”
*Tip*: don’t take pre-med prior to med school (i.e., study engineering, humanities, or something totally unrelated)
Keith’s take on med school and beyond:
- Medical school teaches information as fact
- When talking to people about cancer as a career, he tells them, “look, it’s so simple to find the boundary between what’s known and not known. There’s so much out there we don’t know and in any area of trying to understand physiology, pathophysiology, in the mode of cancer, you will so quickly get to that frontier.”
And then, how do you operate in that frontier?
- Once you’re there, how do you help the patient in front of you, the next wave of patients that are coming in the not too distant future, and then a full generation out?
- Investigation is the only way you can pose yourself as being helpful. And I didn’t get that in medical school, sadly, even though I was at a fantastic environment for learning the archival version of medicine.
Explaining chemotherapy, radiation, and how a cancer develops [23:45]
Defining chemotherapy
- It’s actually not hard to kill a cancer cell, it’s actually very easy to kill a cancer cell:
- Bleach
- Formaldehyde
- “Go to Home Depot, most things on the shelf kill cancer perfectly.”
- Problem is, they kill everything perfectly
- The arbitrage is the following…
{end of show notes preview}
Would you like access to extensive show notes and references for this podcast (and more)?
Check out this post to see an example of what the substantial show notes look like. Become a member today to get access.

Keith Flaherty, M.D.
Keith Flaherty is director of Developmental Therapeutics at the Massachusetts General Hospital Cancer Center and associate professor of medicine at Harvard Medical School. He also serves as editor in chief of the journal, Clinical Cancer Research. He was previously a professor of medicine at the University of Pennsylvania. He is known for his research on targeted therapies for cancer, and in particular for his work on the melanoma drug vemurafenib. In 2013, Massachusetts General Hospital partnered with AstraZeneca to partner Flaherty’s research into developing a formula to identify vulnerabilities of tumors with AstraZeneca’s library of drugs.