Brian Kennedy is a renowned biologist, leader in aging research, and director of the Center for Healthy Longevity at the National University of Singapore. In this episode, Brian shares insights from ongoing human aging studies, including clinical trials of rapamycin and how dosing strategies, timing, and exercise may influence outcomes. He presents two key models of aging—one as a linear accumulation of biological decline and the other as an exponential rise in mortality risk—and explains why traditional models of aging fall short. He also explains why most current aging biomarkers lack clinical utility and describes how his team is working to develop a more actionable biological clock. Additional topics include the potential of compounds like alpha-ketoglutarate, urolithin A, and NAD boosters, along with how lifestyle interventions—such as VO2 max training, strength building, and the use of GLP-1 and SGLT2 drugs—may contribute to longer, healthier lives.
Subscribe on: APPLE PODCASTS | SPOTIFY | RSS | OVERCAST
We discuss:
- Brian’s journey from the Buck Institute to Singapore, and the global evolution of aging research [2:45];
- Rethinking the biology of aging: why models like the hallmarks of aging fall short [9:45];
- How inflammation and mTOR signaling may play a central, causal role in aging [14:15];
- The biological role of mTOR in aging, and the potential of rapamycin to slow aging and enhance immune resilience [17:30];
- Aging as a linear decline in resilience overlaid with non-linear health fluctuations [22:30];
- Speculating on the future of longevity: slowing biological aging through noise reduction and reprogramming [33:30];
- Evaluating the role of the epigenome in aging, and the limits of methylation clocks [39:00];
- Balancing the quest for immortality with the urgent need to improve late-life healthspan [43:00];
- Comparing the big 4 chronic diseases: which are the most inevitable and modifiable? [47:15];
- Exploring potential benefits of rapamycin: how Brian is testing this and other interventions in humans [51:45];
- Testing alpha-ketoglutarate (AKG) for healthspan benefits in aging [1:01:45];
- Exploring urolithin A’s potential to enhance mitochondrial health, reduce frailty, and slow aging [1:05:30];
- The potential of sublingual NAD for longevity, and the combination of NAD and AKG for metabolic and exercise enhancement [1:09:00];
- Other interventions that may promote longevity: spermidine, 17𝛼-estradiol, HRT, and more [1:17:00];
- Biological aging clocks, clinical biomarkers, and a new path to proactive longevity care [1:23:15];
- Evaluating rapamycin, metformin, and GLP-1s for longevity in healthy individuals [1:32:15];
- Why muscle, strength, and fitness are the strongest predictors of healthspan [1:37:30];
- Why combining too many longevity interventions may backfire [1:39:30];
- How increased funding and AI integration could accelerate breakthroughs in aging research [1:41:45];
- The research Brian is most excited about, and the need to balance innovation with safety in longevity clinics [1:47:00];
- Peter’s reflections on emerging interventions and the promise of combining proven aging compounds [1:54:00]; and
- More.
Show Notes
Brian’s journey from the Buck Institute to Singapore, and the global evolution of aging research [2:45]
- Brian used to run an institute called the Buck Institute
Tell folks a little bit about what the Buck is and what you did there
- It was the first institute solely devoted to understanding aging and longevity, and it started around 2000 with some money that was donated by a woman who died in Marin County north of San Francisco
- Brian was the second CEO there in 2010
- There were about 20 faculty at the time all devoted to either aging or aspects of aging, very basic science
- As you can imagine in the 2000s at around 2010, that was a significant component of the aging research field
- It was still a very small field, and so the goal was really to help that institute grow
- It was tough times in the 2010s because the funding levels were low, and it was right before the real interest in aging and longevity happened around 2017, 2018
- So we were really struggling to keep the doors open and the Buck’s doing a lot better now as well as the rest of the aging field
At the time that you were there, how much of the funding came from NIH and how much came from either donations or industry?
- It was heavily oriented to NIH and our goal was to get more industry funding
- We started 7 companies when Brian was there, some of which are still hanging around
- We also really tried to ramp up philanthropy, but philanthropy for aging wasn’t really happening until around 2017, 2018 when people started really getting the idea that you could slow aging and prevent all these diseases and stay healthy and functional
The faculty there are quite the star-studded cast
- Eric Verdin is there (he came after Brian left and took over as CEO)
- Judith Campisi was there until she passed away
- Henry Jasper was there, he’s now gone on to Genentech
- Gordon Lithgow and a bunch of other people working on aging
- It was a good group of people for sure
You alluded to something that happened in 2017, 2018 that brought a lot more interest into the field, what do you think led to this interest?
- You just reach an inflection point, and it’s really hard to know what triggers it
- Calico started a few years before that
- Google’s alphabet companies that was focused on longevity
- They’ve not been very open about what they’re doing, but it triggered a lot of publicity for longevity field
- It got Silicon Valley interested
- Brian and Matt used this slide that you have aging pointing to all these different diseases [shown below]
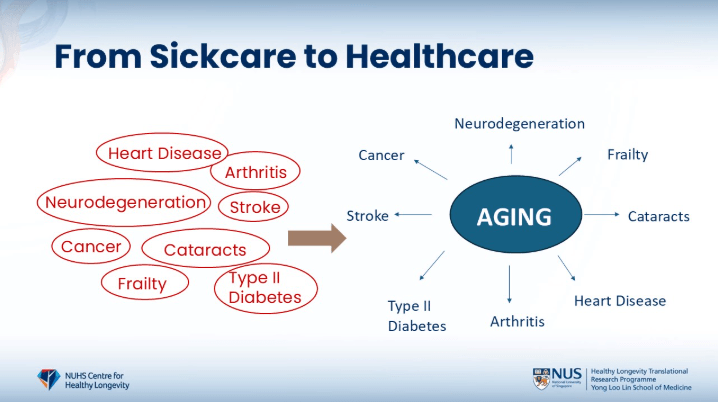
Figure 1. Diseases related to aging. Image credit: Brian Kennedy
Brian explains, “We started using that slide around 2005, and we were making the point around healthspan shortly afterwards and really this idea of preventing disease and keeping people healthy, interacting earlier.”
- Now Brian is so sick of that slide; he can’t look at it anymore
- Back then, there were a few scientists doing it and it finally helped trigger a movement (hopefully)
Do you think the field would’ve accelerated sooner had it not been for some notable setbacks?
- For example, resveratrol
- Peter doesn’t remember exactly when GSK bought resveratrol, around 2006, 2007
- By about 2010, it was clear to him that was not going to work
- GSK probably knew sooner
Do you think that that type of hype with nothing to show for it was a negative force in that equation and maybe this inflection point could have happened sooner,and it could have been easier 7 raised funds if there had been less of those examples?
- It’s hard to know
- It’s unfortunate that investors made money off that deal when what was developed wasn’t going anywhere
It probably slowed things down a little bit because there’s always this doubt about whether you can slow the aging process
- When you have a major effort that’s triggered around trying to do that, even though they ended up focusing on disease, and when something like that fails, it probably does slow down other investor interests
- We can talk about the struggles of longevity biotech companies in that way
Today you’re in Singapore; tell me what you’re doing there
- Brian has one foot in academics and one foot in the private sector these days
- On the academic side, he’s really focused on targeting aging
- And that comes back to what he alluded to with the biotech companies a minute ago: a lot of them are targeting aging pathways, but to raise money and get their drugs tested, they have to turn to some disease indication (which is understandable)
- The companies he’s involved with do that too, but that’s not what we really want to do
“What we really want to do is slow the aging process and keep you from getting sick.”‒ Brian Kennedy
- In an academic setting, we can test that clinically
- We basically have a whole range of animal models, a pipeline from yeast, worms, flies, killifish, mice, and humans (billion years of evolution there)
- We bring interventions in at the right place, validate them, really believing the idea that if it works across different model organisms, it’s more likely to work in humans
- Then we design human clinical intervention studies to validate that they’re targeting the aging process
Brian adds, “I don’t think anybody knows exactly how to do that yet (including us), but we’re doing our best and learning as we go.”
How is the institute you’re at funded?
- [Brian is at the Centre for Healthy Longevity at National University in Singapore]
- Mostly through the university and the government in Singapore, but we also have some philanthropy, and we do contract sponsored research to test interventions from companies sometimes
How many PIs are there?
- Brian runs a program that has about 35 PIs in it
- But a lot of them are doing other things
- They’re not all focused on that one concept he just spoke about
- They have their projects around Alzheimer’s disease
- Or we just have this guy Michael Chee, who’s working on sleep and aging, which is so understudied
- It’s kind of like an academic department where people have their own projects they’re focusing on
Is there any department at a US university that brings together as many people that are focused on specifics of aging this way?
- Brian doesn’t think so, although it depends on how you define aging
- If you branch it out to cancer then there are, but he’s not sure if that is focused on the actual aging process
Rethinking the biology of aging: why models like the hallmarks of aging fall short [9:45]
- Peter wants to start with a question that he thinks they’ll end up coming back to because it’s so fundamental
Peter adds, “I enjoy going down the rabbit hole of fundamental questions in physics.”
What do we think is actually causing aging?
{end of show notes preview}
Would you like access to extensive show notes and references for this podcast (and more)?
Check out this post to see an example of what the substantial show notes look like. Become a member today to get access.

Brian Kennedy, Ph.D.
Brian Kennedy earned his bachelor’s degree in biochemistry and mathematics from Northwestern University. He earned his Ph.D. in biology at MIT and completed postdoctoral research at Massachusetts General Hospital Cancer Center. Dr. Kennedy was a professor of Biochemistry at the University of Washington in Seattle until 2010. He was a visiting professor at Guangdong Medical College in China until 2014. He served as the President and CEO of the Buck Institute for Research on Ageing from 2010-2016. Currently, Dr. Kennedy is a distinguished professor of Biochemistry and Physiology at the Yon Loo Lin School of Medicine at the National University of Singapore (NUS) where he also serves as: Director of the Center for Healthy Longevity, Programme Director of the Healthy Longevity Translational Research Programme, and Director of the Asian Centre for Reproductive Longevity and Equality. His affiliations include Adjunct Senior Principal Investigator at the Singapore Institute for Clinical Sciences at A*STAR; and Adjunct Professor at the Leonard Davis School of Gerontology at USC in Los Angeles. Dr. Kennedy’s research focuses on understanding the biology of aging and translating research discoveries into new ways of delaying, detecting, preventing, and treating human aging and associated diseases. He uses multiple model organisms for these purposes, relying on non-vertebrates for discovery-based approaches to generate hypotheses regarding aging mechanisms and studies in mammals to test hypotheses and to develop translational strategies. He then conducts clinical studies in humans to evaluate these strategies and interventions that target aging. [NUS]




