Check out more content with expert, Matt Kaeberlein, Ph.D.:
- (Aug 20, 2018) Rapamycin and dogs — man’s best friends? — living longer, healthier lives and turning back the clock on aging and age-related diseases
- (Sep 13, 2021) The biology of aging, rapamycin, and other interventions that target the aging process
- (May 16, 2022) AMA #35: “Anti-Aging” Drugs — NAD+, metformin, & rapamycin
Matt Kaeberlein is globally recognized for his research on the biology of aging and is a previous guest on The Drive. In this episode, Matt defines aging, the relationship between aging, chronic inflammation, and the immune system, and talks extensively about the most exciting molecules for extending lifespan. He discusses the current state of the literature of testing rapamycin (and rapalogs) in animals and humans, including Matt’s Dog Aging Project, and provides insights into how we can improve future trials by conceptualizing risk, choosing better endpoints, and working with regulators to approve such trials. He also examines the connection between aging and periodontal disease, biomarkers of aging, and epigenetic clocks. Finally, they explore some of the biological pathways involved in aging, including mTOR and its complexes, sirtuins, NAD, and NAD precursors.
Subscribe on: APPLE PODCASTS | RSS | GOOGLE | OVERCAST | STITCHER
We discuss:
- The various definitions of aging [3:25];
- The relationship between disease and the biology of aging [16:15];
- Potential for lifespan extension when targeting diseases compared to targeting biological aging [22:45];
- Rapamycin as a longevity agent and the challenges of targeting the biology of aging with molecules [32:45];
- Human studies using rapalogs for enhanced immune function [39:30];
- The role of inflammation in functional declines and diseases of aging [50:45];
- Study showing rapalogs may improve the immune response to a vaccine [56:15];
- Roadblocks to studying gero-protective molecules in humans [1:01:30];
- Potential benefits of rapamycin for age-related diseases—periodontal, reproductive function, and more [1:12:15];
- Debating the ideal length and frequency of rapamycin treatment for various indications like inflammation and longevity [1:21:30];
- Biomarkers of aging and epigenetic clocks [1:29:15];
- Prospects of a test that could calculate biological age [1:37:45];
- The Dog Aging Project testing rapamycin in pet dogs [1:42:30];
- The role of the mTOR complexes [1:58:30];
- mTor inhibitor called Torin2, mitochondrial disease and other potential pathways [2:09:45];
- Catalytic inhibitors, sirtuins, and NAD [2:19:15];
- NAD precursors: help or hype? [2:28:15]; and
- More.
Get Peter’s expertise in your inbox 100% free.
Sign up to receive An Introductory Guide to Longevity by Peter Attia, weekly longevity-focused articles, and new podcast announcements.The various definitions of aging [3:25]
- Last time Matt was on the podcast, they talked about rapamycin and the mammalian target of rapamycin (mTOR) pathway
- Peter finds Matt to be very thoughtful across longevity topics, so wants to have an even broader discussion today
- What’s the best way to define aging?
- It varies depending on the situation
- Can be molecular: the types of molecular damage and their consequences
- mitochondrial dysfunction, telomere shortening, cellular senescence, etc. all happen at the cellular and molecular level and contribute to functional decline and diseases
- Matt has begun to develop a greater appreciation for a functional definition of aging
- There are functional declines across every organ system in the body
- Things like frailty are an important component of defining aging
- Other aspects of aging, like the social component, intersect with the biological part and are as important to quality of life
- Over time, scientists have established the nine hallmarks of aging [see special supplementary section for details]
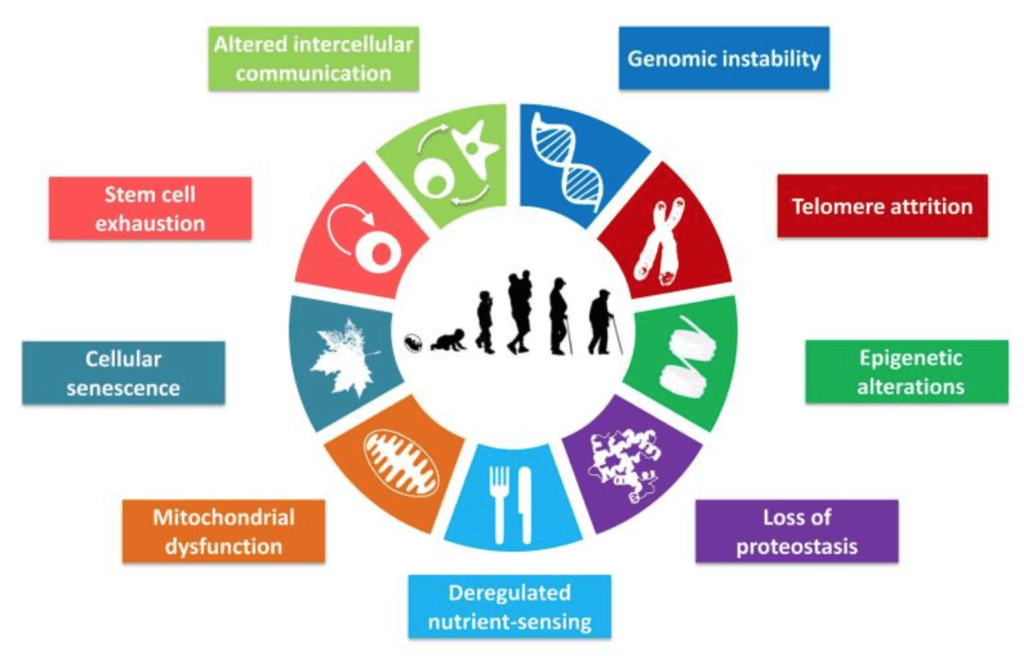
Figure 1. The Nine Hallmarks of Aging. Image credit: López-Otín et al. 2013
- There are also outward expressions of these processes: frailty, diseases like cancer, cardiovascular disease, and dementia
- Peter asks if disease is the link between cellular and phenotypic declines
- Matt says he thinks of it the other way around: the functional declines often precede disease and are as (or more) important from a quality of life perspective as we get older
- These functional declines happen in every organ and tissue, often long before one is diagnosed with any disease
- At 50, Matt has not been diagnosed with any age-related diseases, but he has a lot of functional declines compared to his 25-year-old self
- Functional declines are often the first observable signs of aging
- Two points:
- we really don’t have a good understanding of when the pathology of the disease is no longer normative aging
- we have some understanding of the molecular cellular mechanisms that drive biological aging and contribute to our risk of developing age-related diseases that are major causes of death and disability
- But at some point, disease pathology is not necessarily at a molecular mechanistic level and becomes something different
- There’s not yet a consensus whether rapamycin affects biological aging
“It’s not clear that the same intervention [that affects biological aging] is going to be effective once a pathology progresses to the point that it’s not the same mechanism anymore. So I think that’s a really important point that sometimes gets lost in this discussion of aging and disease.” —Matt Kaeberlein
Peter asks how this point can be illustrated with one of the “big three” age-related diseases: atherosclerosis, cancer, and dementia
- Matt says cancer provides a good illustration of the concept
- mTOR, the protein that rapamycin inhibits, plays a fundamental role in regulating cell division and cell cycle
- if you inhibit mTOR in a non-cancerous cell enough, you will stop the cell cycle
- but some cancer-causing mutations will not respond to the mTOR inhibition
- so rapamycin will be quite effective at preventing cancer before a mutation happens, but afterwards the cell will not respond to rapamycin anymore an the cell cycle will not be affected
- the mechanism has changed, so the mechanisms that are important for preventing cancer before that mutation occurred are different from the mechanism that might halt the growth of that cancer after the mutation has occurred
- Peter says it’s similar to nutrition: as far as there is an optimal nutrition program to prevent a condition, it might not be the same as the optimal nutritional strategy to treat the disease once it develops
- Peter thinks a ketogenic diet is probably the best treatment for someone with type 2 diabetes, which is essentially a carbohydrate intolerance disorder
- But one likely does not need to be on a ketogenic diet to prevent diabetes
Normative aging vs. disease [14:30]
- Cynthia Kenyon has observed that using a disease-based definition for aging is not quite the right framework because it’s not clear at what point a disease is a disease
- If everyone who lives to a certain age has cancer, it’s become normative aging rather than a disease
- 0.0004% of people live to 100 because they did not succumb to a disease – what does that say about their normative aging process vs. everyone else’s?
- Peter reflects, “I literally still don’t think I understand what aging is”
“I don’t think I will ever understand aging fully. And I don’t think the field will. … But I also believe that we don’t have to understand it fully to be able to have an impact on the biology of aging through interventions. … I feel like I’ve got a conceptual flavor for what aging is … and enough information that I can come up with rational approaches to target those mechanisms … that should have an impact on health and longevity. … And then we have to test those predictions.” —Matt Kaeberlein
The relationship between disease and the biology of aging [16:15]
- The relationship between disease and the biology of aging is underappreciated
- Matt notes that people often debate whether the biology of aging (the molecular hallmarks) cause diseases like Alzheimer’s disease, cancer, and cardiovascular disease
- Matt thinks the data show that they do make a causal contribution to disease, but he also thinks “it just doesn’t matter whether aging causes disease or creates a permissive physiological state for disease”
{end of show notes preview}
Would you like access to extensive show notes and references for this podcast (and more)?
Check out this post to see an example of what the substantial show notes look like. Become a member today to get access.

Matt Kaeberlein, Ph.D.
Dr. Matt Kaeberlein is a Professor of Pathology, Adjunct Professor of Genome Sciences, and Adjunct Professor of Oral Health Sciences at the University of Washington. His research interests are focused on basic mechanisms of aging in order to facilitate translational interventions that promote healthspan and improve quality of life. He has published nearly 200 papers in top scientific journals and has been recognized by several prestigious awards, including a Breakthroughs in Gerontology Award, an Alzheimer’s Association Young Investigator Award, an Ellison Medical Foundation New Scholar in Aging Award, a Murdock Trust Award, a Pioneer in Aging Award, and the Vincent Cristofalo Rising Star in Aging Research. His contributions have also been recognized with Fellow status in the American Association for the Advancement of Science, the American Aging Association, and the Gerontological Society of America. Dr. Kaeberlein is a past President of the American Aging Association and has served on their Executive Committee and Board of Directors since 2012. He has also served as a member of the Board of Directors for the Federation of American Societies for Experimental Biology and is currently the Chair of the Biological Sciences Section of the Gerontological Society of America. Dr. Kaeberlein serves on the editorial boards for several journals, including Science and eLife. Dr. Kaeberlein’s scientific discoveries have generated substantial public interest, with featured stories in major media outlets including appearing on the front page of the New York Times, the Today Show, CNN, the UK Telegraph, Popular Science, Time Magazine,
Scientific American, NPR, USA Today, National Geographic, and many others. In addition to his primary appointments, Dr. Kaeberlein is the co-Director of the University of Washington Nathan Shock Center of Excellence in the Basic Biology of Aging, the founding Director of the Healthy Aging and Longevity Research Institute at the University of Washington, and founder and co-Director of the Dog Aging Project. [kaeberleinlab.org]
Matt on Facebook: Matt Kaeberlein
Matt on Instagram: @mkaeberlein
Matt on Twitter: @mkaeberlein



