Prostate cancer is the second leading cause of cancer death in men. Only lung cancer claims more lives. It’s one of those unfortunate diseases that will almost certainly touch every one of us, if not personally, then through someone we love. A staggering 12.8% of men, 1 in 8, will be diagnosed with prostate cancer in their life, and 36,000 men are expected to die from prostate cancer in the United States this year alone.1
It is fortunately one of those diseases for which we have incredibly powerful treatment options—the 15-year survival rate from first diagnosis pushes 97%. It’s also a disease for which we have incredibly simple and increasingly effective early detection strategies.
Yet we’re facing a conundrum: Despite powerful early detection tools, data from the USA and Canada show that advanced stage prostate cancers are on the rise.1,2 And the gains we were making in mortality have all but flatlined across the board. Some even reversed.
This begs the question: Why? Why are advances in our therapeutic strategies not leading to better outcomes? Why, with broadly accessible screening tools, are we catching tumors only after they’ve developed into advanced metastatic disease? And what can we do?
Prostate-specific antigen (PSA)
First available in the early 90s, the standard blood assay to detect prostate malignancies measures prostate-specific antigen (PSA). PSA is a protein produced by the prostate to aid in maintaining the liquidity of seminal fluid. A small amount of PSA leaks through prostate vasculature into the blood, which can then be quantified in a standard blood draw.
As the prostate increases in size, as it will in most men as they age, the amount of PSA detectable in the blood proportionally increases. The initial application of the PSA test held that abnormally high levels of PSA in the blood therefore indicated an abnormally large prostate, potentially implicating malignant growth. A high reading on a PSA test would lead to an ultrasound, a positive signal here would lead to a biopsy, and a biopsy detecting any cancerous growth would lead to treatment.
In trials run over 14 and 16 years,3,4 routine PSA screening was found to reduce prostate cancer mortality by 44% and 64%, respectively—preventing as many as 3.1 cases of metastatic (i.e., normally fatal) disease for every 1000 men screened.5
This test is dirt cheap to run and is covered 100% by most insurances.
So why not test for PSA?
The pipeline leading from elevated PSA levels directly to ultrasound and biopsy became a concern—false alarms were too common. While some false alarms are worth the trouble of ruling out cancer, the signal-to-noise ratio and burden of biopsy were particularly high. PSA levels outside of the normal range are not always indicative of pathology, but prostate biopsies are always invasive.
The most common method for sampling prostate tissue is through the rectum wall. Prophylactic enema and antibiotics are useful for minimizing infection from the procedure, but the reality is you are puncturing through a bacteria-rich rectum and creating a contaminated tract into the otherwise sterile prostate. Up to 5-7% of these biopsies result in an infection.6 Prostate infections are serious, particularly if they spread to the bloodstream, and often lead to emergency room visits, hospital stays, and time out of work.
Beyond the physical cost of biopsy, the psychological and physical impact of cancer diagnosis and treatment can be enormous. In the 1990s and early 2000s, a cancer detected almost always resulted in a cancer treatment. But we now know that many of the cancers detected in the early PSA screening-era were indolent, and would therefore not require any treatment.
Given these costs, the United States Preventative Services Task Force (USPSTF) guideline panel ceased recommending PSA screening. In 2008 they ended recommendations for PSA testing in men over 70 years of age, ultimately recommending against screening for men of all ages in 2012.7
The data that the USPSTF cited to support this change came from a US-led trial called the Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial, or PLCO, which showed that screening saved at most 1.3 lives per 1000 men screened. In addition to the 5–7% chance of infection from biopsy, treatment side effects can be rather severe. They specifically cited erectile dysfunction from surgery, radiation, and hormone therapies; urinary incontinence from radiation and surgery; and loss of bowel control from radiation.
The USPSTF’s recommendation boiled down to this logic: saving 0.1% of lives while causing major complications, and in rare cases even death, in a meaningful proportion of other men isn’t worth it. The risks of overdiagnosis and overtreatment did not outweigh the benefits.
This motivation is at least defensible, if not noble to our utilitarian sensibilities. But it has been nearly 15 years. If the USPSTF’s recommendations were built on a solid foundation, we would expect no more than a 0.1% change in high grade or metastatic prostate cancer, or prostate cancer deaths in the absence of PSA screening.
So we have a responsibility to ask—how has the change in recommendations actually panned out? And with 15 years of advancements under our belts, do the risks still outweigh the benefits?
Our research team spends hundreds of hours each month vetting studies and distilling dense literature to deliver evidence-informed insights on health and longevity. If you find value in our work, consider becoming a premium member and supporting our mission.











Metastatic prostate cancer on the rise
Lumping together all prostate cancers diagnosed at stages I, II, or III, there is a >99% 5 year survival from time of diagnosis. This drops to 38% when diagnosed at stage IV.1 For stage IV, median survival time is 30 months.8
When prostate cancer is diagnosed is critical to survival.
Data published in the last 6 months by the USA and Canada build on a previous report from 2016, with both recent studies showing that more and more cases are being caught only once disease has become metastatic. Metastasis is the hallmark of stage IV disease, where tumors are spreading throughout the body. At this point, prostate cancer is largely incurable.
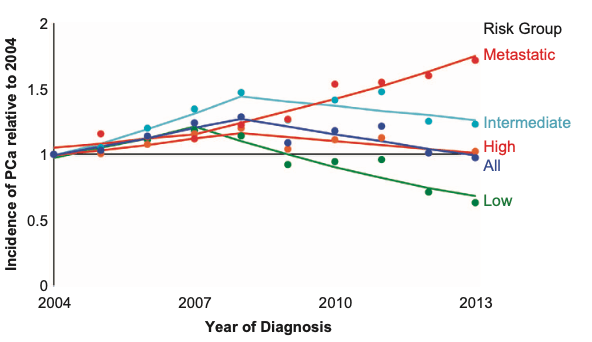
Scientists in the field suspected that the cultural shift away from screening, first initiated by the USPSTF in 2008, risked exposing older men to higher rates of advanced stage cancers. The latest data don’t only show this to be true, they also find an even more problematic trend—we’re finding more advanced stage cancer in young men too. In Canada, there has been a 3.7% year-over-year increase in stage IV diagnoses in men under 75 from 2010-2017, despite declining numbers of total cancers detected at this age range.2
Similarly, the picture from the USA1 is exceedingly clear. Since recommendations shifted in 2012, there has been a 0.1% decrease in stage II cancers detected year-over-year in all ages. This has been accompanied by a 3.3% yearly increase in stage III cancers and a 6.0% yearly increase in stage IV. These increases in stages III and IV far outpace the 0.8% increase in total number of cases detected.
In other words: We are finding disease too late.
This outcome is not because prostate cancer incidence itself is rising—indeed, we can even see the opposite in some age ranges—it is that we have stopped looking for early-stage disease. We are increasingly finding cancers only once they become symptomatic, often at stages III and IV. And stage IV diagnoses drive nearly all instances of prostate cancer deaths.
If you stop looking for early stage cancer, you will only find advanced stage disease.
We find ourselves going forwards and backwards at the same time. As stated by the authors in Canada, “New life-prolonging treatments for patients with metastatic disease have increased the survival for stage IV cancer. However, overall survival decreased from when screening was recommended, attributable to the increased number of incurable cases.”
Fortunately, this does not have to be the case.
Screening smarter and harder—refining PSA screening
The USPSTF’s guidelines were established when the tools surrounding PSA were less sophisticated. The pipeline was rather direct: high PSA led to ultrasound, an enlarged prostate seen by ultrasound led to biopsy, detection of any cancer led to treatment. The decision algorithm around PSA testing, imaging, biopsying, and treatment has since become significantly more nuanced.
PSA testing is best utilized as a routine monitor—not an absolute value. Individual data reads are not terribly informative, varying naturally by 15% day-to-day10 and jumping acutely by as much as 40% based on recent prostate activity (e.g., ejaculation).11 There is also enormous person-to-person variability: while the median PSA value for a healthy 60-69 year old man is 1.0–1.2 ng/mL, the 95th percentile for the same age range goes as high as 4.9 ng/mL, even in the absence of prostate cancer (see below).
Table: Median PSA by age in men without prostate cancer (95th percentile)12
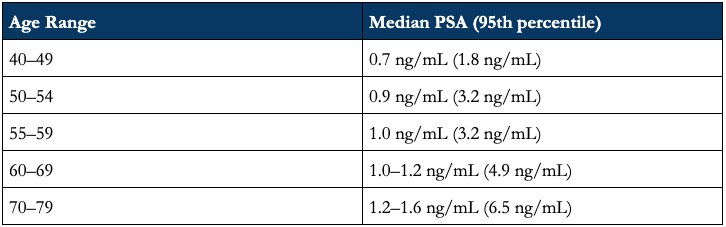
Routine testing helps us peak through the noise. (And again, it’s dirt cheap.)
By mapping an individual patient’s baseline then tracking the rate of change through time, we get a value known as PSA Velocity. While the prostate does typically enlarge throughout life, rapid PSA increases can indicate a pathological enlargement of the prostate, increased leaking of PSA into the blood, or both. Each of which could indicate malignant growth.
These patient-specific trends allow us to avoid overreactions to individual deviations from population norms: abnormal increases through time serve as the red flag. This is precision medicine for pennies—PSA velocity is tuned to the unique individual, normalized against their own baseline and trends.
As an example, a 55 year old patient with a PSA of 1.3 ng/mL would not be flagged, despite being above the median for their age. Should we see increases in excess of 0.35 ng/mL/year over an 18 month period, however, we would follow up with further testing. If that same patient has a PSA value of 1.7 one year later, then 1.9 six month after that—an increase of 0.6 ng/mL over 18 months, at a rate of 0.4 ng/mL/year—this sustained rate of increase is our first red flag. (Worth noting: for patients with baseline PSA values above 4.0, the velocity cut off increases to 0.75 ng/mL/year.)
That said, even using PSA velocity, PSA screening alone is no longer sufficient to immediately move to ultrasound and biopsy—MRI now serves as a less invasive, higher resolution intermediary.
The 3D map provided by MRI permits us to calculate PSA density as well. PSA density is a simple calculation: the concentration of PSA in the blood divided by the volume of the prostate.
PSA levels in the blood will rise as the prostate increases in size throughout life. While volume and PSA concentration tend to increase proportionally in a healthy male, disruptions to the endothelium or underlying architecture can permit increased flow of PSA from the prostate into the bloodstream. Disruption of the prostate gland’s architecture, or the phenomenon of “leaky” blood vessels, are each hallmarks of malignant growth.
PSA density thereby serves as yet another metric to assess cancer risk before a biopsy even enters the equation: If PSA levels in the blood are increasing significantly faster than the size of the prostate, it implicates increased PSA leakage from the prostate into the blood. This leakage implicates disruption of normal prostate morphology and/or blood vessel barriers, and either kind of disruption implicates cancerous growth. This gives us a strong signal well before needing to biopsy.
With PSA velocity from repeat tests and PSA density from MRI imaging, the insights leverageable by PSA testing have dramatically improved in recent decades. But PSA testing is rarely utilized alone—it is just the first signal. This combination of PSA testing and MRI has become increasingly valued pre-biopsy, and is poised to become even more common and more accessible in the immediate future.
MRI accessibility
In addition to PSA density, the greatest utility of MRI lies in the identification of lesions or suspicious regions without invasive probing. It can meaningfully rule out or rule in the need to biopsy, and further guide any necessary biopsies to the regions most likely to hand us a smoking gun.
It was previously burdensome, and for many patients outright infeasible, to conduct high quality MRIs for detection prior to biopsy. The MRIs utilized gadolinium contrast—increasing costs for the patient, necessitating a medical practitioner to administer the injection, and requiring more time on the scanner for each MRI. This limits the number of possible patients and adds meaningful time and personnel strains to the healthcare system. But a recent study is likely to shift standard practice.
The PRIME trial,13 published in 2025, compared the gold standard “multiparametric” MRI (T2-weighted, Diffusion-weighted, and dynamic contrast-enhanced imaging) to a simpler, contrast-free “biparametric” MRI (only T2- and Diffusion-weighted imaging) for the detection of prostate cancer. They set out to establish noninferiority of the simpler method—that is, they wanted to assess if the simpler biparametric method was statistically worse than or equivalent to the contrast-enhanced, multiparametric gold standard.
The PRIME trial found that the contrast-free, biparametric MRI was noninferior to the gold standard. Biparametric MRI detected clinically significant prostate cancer in 143 of 490 patients flagged by PSA screening, while the gold-standard multiparametric MRI detected clinically significant cancer in 145 of the same 490 patients. This yielded an insignificant difference of -0.4 percentage points between methods (29.2% vs 29.6%; 95% CI: -1.2–0.4).
More patients can benefit from diagnostic MRI when the cost is reduced. The contrast-free MRI takes half as much time on the scanner (15–20 vs 30–40 minutes), increasing throughput on already available resources. And the lack of contrast alleviates healthcare system burden, removing the need for patient monitoring by a medical practitioner throughout the scan.
MRI has been increasingly recognized as a valuable tool in assessing prostate health without biopsy—we now know we can dramatically increase accessibility while trimming time and technology investment, all without reducing quality of care.
Cleaning up the biopsy
If the MRI suggests a biopsy is necessary, we’ve also made strides towards improving both the safety and accuracy of our biopsying technique. The most common form of biopsy is still transrectal (TR)—running that risk of introducing bacteria from the digestive tract into the prostate. It also biases sampling to the rear of the prostate, near the rectum, and can miss the front and base of the prostate. By entering through the skin between the anus and scrotum, transperineal (TP) prostate biopsy circumvents the rectum, reducing the risk of infection while increasing access to the prostate’s base and front.
A multi-center phase 3 trial with over 1700 patients, conducted by Ted Schaeffer and colleagues, compared cancer detection and complications after either TR or TP biopsies. They found TP biopsy increased their detection of high grade cancers while decreasing detection of low grade cancers, suggesting the transperineal approach was more sensitive for correctly identifying aggressive tumors. They also report that not a single patient undergoing TP biopsy developed an infection in this study.14
This method is not yet performed everywhere—only 37.2% of USA Urologists report performing TP biopsies in a recent census, compared to 99.4% performing TR15—but it demonstrates meaningful progress towards minimizing the harms and maximizing the utility of biopsying after MRI. TP biopsy does not require antibiotics, simplifying the process for the patient and preserving antibiotic use for when it’s truly needed. Even without antibiotics, something near 0% of patients will develop infections. And improved accuracy gives us a clearer picture, improving our confidence in any treatment approach that could follow.
Avoiding overtreatment
Not all prostate cancers are ultimately fatal. While our treatments are effective, they can still carry side effects we want to avoid if at all possible.
Patients who go through the entire above process can find a tumor but still not require immediate treatment. Pathologists will analyze the biopsy, then grade the cancerous tissue on a scale determined by how normal-to-aggressive the tumor cells look, relative to healthy prostate tissue. They will be assigned a Grade Group (GG) 1–5.
A score of GG3, 4, or 5 indicates increasing proportions of increasingly malignant cells. This will typically lead to immediate treatment—we know these are the dangerous tumors.
Scores of GG1 and GG2 are considered sufficiently low-risk that treatment is often not yet necessary. These patients are then put into active surveillance, but no treatment is initiated until further red flags are raised.
These low grade tumors still risk developing into more severe cancer, even if not immediately dangerous. Active monitoring can entail enhanced frequency of PSA testing, additional blood tests like PHI/4Kscore to monitor prostate antigens beyond typical PSA, tracking of symptoms related to difficulty or pain with urination, and follow up imaging to track growth.
If warning signs show up during this active monitoring, a follow up biopsy may be performed, ensuring that timely treatment occurs once it is required. But only once it is required.
Taken together, with the significantly more sophisticated tools and risk assessments available today, we can delay, or in some patients outright avoid, the burden of aggressive cancer treatment.
The PLCO was fatally flawed
We just spent some considerable time taking the USPSTF’s recommendations seriously. The process of screening, imaging, biopsying, and treating cancer can take a real toll on people. Being diagnosed with cancer, of any kind, presents a real psychological burden. Imaging and biopsying aren’t completely cost-less no matter how you frame it, and treatment side effects are effectively unavoidable.
That said, there was a monumental failure in the formulation of these guidelines. Urologists have known for years that the data used by the USPSTF was opaquely, fundamentally, and fatally flawed. The USPSTF’s claim that screening saves at most 1.3 lives per 1000 men screened is built on a study so irresponsibly reported that it is borderline unbelievable it passed the peer review process, let alone review and utilization by a panel of experts tasked with guiding the health of the American public.
The USPSTF primarily informed its decisions on screening efficacy based on two large studies, favoring the one conducted in the United States—the PLCO mentioned earlier. In the prostate screening section of this study, they randomized participants to groups either undergoing PSA screening (intervention arm) or not undergoing PSA screening (control arm). They found virtually no difference between these groups in prostate cancer mortality, supporting their claim that no more than 0.1% of men screened will survive their cancer as a result of PSA testing.What they didn’t account for when reporting differences between arms in this study was, unbelievably, that both groups underwent PSA screening. The study authors may not have provided the PSA tests, but surveys of the participants showed that 39–64% of people in the “no screening” control arm reported having a PSA test “within the past year” every single year of the study. 60–84% in the “no screening” arm reported a test within the last 3 years, again, every single year of the study.16
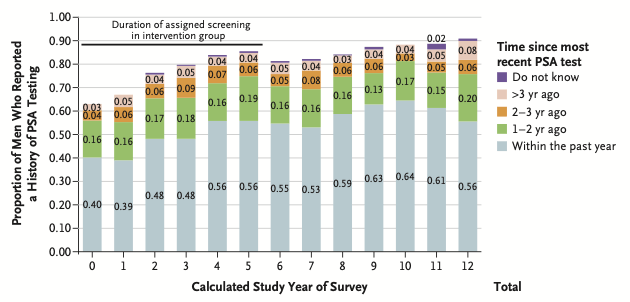
These data were made widely available in 2016, 4 and 8 years after the USPSTF’s recommendations. By the final year of survey responses, 90% of people in the “no PSA screening” group reported having undergone at least one PSA test in the past. Many years, these surveys even showed more participants reported PSA testing within the last year in the control arm than in the group supposedly undergoing regular screening.
Given the enormous contamination of the no screening group, in 2017, separate analyses that mathematically accounted for frequency of screening within these data allowed researchers to still investigate the impact of PSA testing on prostate cancer mortality. They calculated a mean lead time (MLT) to describe how much earlier cancers were able to be caught as a result of screening, using this calculation to impute a reduction in mortality as a function of diagnosis earlier in disease progression. Performed this way, the same PLCO datathat were used to recommend against PSA testing then show a 27-32% reduction in prostate cancer mortality as a function of regular PSA testing.17
The last USPSTF recommendation came in 2018, still not directly recommending PSA testing for men 55-69, and still recommending against it for men 70+. Yet it is clear that 27-32% of prostate cancer deaths that evolved without PSA detection could have been prevented by an honest analysis of the PLCO data.
It is time for the USPSTF to update its recommendations. The homologous organization in Canada, the Canadian Task Force on Preventive Health Care (CTFPHC) should follow suit. But until they do, it is time for us as clinicians and patients to take PSA testing seriously on our own.
Eradicating prostate cancer deaths
We began with the observation that the 2008 and 2012 recommendations against PSA testing led to a demonstrable increase in the incidence of incurable cancers.
In 2026, some of the facts haven’t changed. PSA testing as a single value, taken once every few years or more, is still an insufficient tool for early cancer detection. The test is too noisy with too much individual variation, even between healthy prostates. What has changed is how PSA can be put into practice.
PSA testing is dirt cheap, and regular testing allows us to tune values to the unique patient and avoid overreactions to individual variations. PSA’s utility improves substantially when interpreted in context—PSA velocity, PSA density, and a judicious use of MRI meaningfully enhance risk stratification and reduce unnecessary invasive procedures.
We’re making progress in the safety and efficacy of prostate biopsies. And we now only recommend treatment in response to demonstrably higher risk tumor pathologies.
We can combine data from multiple sources when making treatment decisions, minimizing the harms of overtreatment while avoiding the harms of just closing our eyes to the problem.
So what should we do?
Prostate cancer is the second leading cause of cancer death in men and is not being afforded the urgency it is owed. It is frankly unconscionable that we still catch these cancers too late to save people’s lives, especially given the pattern of most prostate cancers’ development.
Cancers can broadly be divided into two categories: those that progress in a relatively predictable, stepwise fashion from non-malignant to malignant states, and those that do not. Prostate cancer falls into the first category; pancreatic and many forms of breast cancer fall into the latter.
This distinction matters. For cancers that do not follow a predictable, screen-detectable precursor pathway, even aggressive screening will not reliably prevent all metastatic disease. That biological reality makes them frustrating. But it is precisely the opposite dynamic that makes deaths from prostate cancer so tragic: When screened appropriately, these cancers can often be detected before metastatic potential is realized. The fact that prostate cancer remains the second leading cause of cancer death in men underscores a failure of implementation, rather than a cruelty of biology alone.
By comparison, if we were lucky enough to have a tool for breast cancer detection costing less than a cup of coffee, easily integrated into routine blood work, which regularly flagged cancer years before metastasizing… in what world would we not have this in every doctor’s office, in every doctor’s hands?
For prostate cancer, we have these tools at our disposal. Your primary care physician can add PSA testing to your bloodwork any time they do a draw. It may even be there already. Our problem is not the absence of tools, but insufficient application and education in how to use them correctly.
We’ve seen this in our clinic (A summary of how we use PSA testing in our practice can be found in this past newsletter). Patients who already have PSA testing from other providers come to our practice and we calculate their PSA velocity. With immediate MRI and biopsy, we then catch prostate cancers that would have almost certainly become metastatic in the next one to two years.
Cancers were missed because of a lack of awareness. In some of these patients, the red flags were lying in data already in hand.
Vigilance during DHT suppression
This brings us to an important, often-missed complication: The use of 5-alpha reductase inhibitors (5-ARI)—like finasteride—has skyrocketed in recent years for the treatment or prevention of hair loss. We’ve talked at some length about the potential risks of finasteride in the development of prostate cancer and we will have more content diving into these data. For now, suffice it to say that we do not believe there is particularly strong data to suggest that finasteride causes prostate cancer de novo. What there is, critically, is a clear demonstration that finasteride interferes with PSA’s ability to flag early stage prostate cancer if we don’t know what we’re supposed to be looking for.
5-ARIs inhibit the conversion of testosterone to the more potent androgen dihydrotestosterone (DHT). In the prostate, inhibition of DHT production also dramatically reduces the production of PSA. A good rule of thumb for finasteride is to expect that PSA values will be cut by one third over 6 months, cut in half by a year, with this suppressed PSA value being maintained as long as 5-ARI therapy is maintained. I will restate this for emphasis: A PSA value obtained while on finasteride for 12 months or longer needs to be doubled, at least, to be accurately understood.
5-ARI-mediated suppression of PSA increases the longer patients are on therapy—in our hands, we multiply the PSA value by 2x for years 1–2, multiply by 2.3x for years 2–7, and multiply by 2.5x for years 7+. While the FDA label recommends multiplying by 2, we do this based on data from the Prostate Cancer Prevention Trial18 performed in the late 90s and early 2000s, as well as a follow up analysis performed in 2005 by Ruth Etzioni and colleagues.19
Looking at PSA velocity, however, we can take a simpler approach: After 12 months, we should not expect PSA values to rise any longer. The prostate will still increase in size as the patient ages. However, the cumulatively increasing suppression of PSA should nearly-negate tissue growth’s effect on PSA values. In a study of 19,000 older men, of patients who had a prostate biopsy negative for cancer at the end of the 7 year study period, those taking finasteride had an average 2% annual decrease in PSA levels.19
This effect is not widely known by general practitioners. In a retrospective cohort of Veteran’s Affairs patients undergoing PSA screening, men taking finasteride were diagnosed with metastatic prostate cancer more than twice as often as patients not taking a 5-ARI (6.7% vs 2.9%). The physicians appear to have been unaware of the severe suppression of PSA by 5-ARIs, ultimately missing that any raw values or velocity increases should have been at least doubled in the 5-ARI patients. Between patients taking finasteride or not, the groups had similar PSA levels at cancer diagnosis: 6.8 vs 6.4 ng/mL. But when the study authors multiplied values to account for PSA suppression by 5-ARI medications, they found this difference would instead have been 13.5 ng/mL vs 6.4 ng/mL.20
Like our USPSTF guidelines recommending against PSA testing, here, even in patients undergoing PSA screens, insufficient physician knowledge surrounding the tool had permitted prostate malignancies to grow unchecked.
We find this point particularly important because we know many men are taking finasteride. But physicians prescribe these drugs without understanding their effect on prostate cancer screening. And many people fail to inform their general practitioner that they receive a finasteride prescription from an online clinic, inadvertently misleading even an informed practitioner about how to interpret PSA testing values.
For those of you taking finasteride, the onus may fall on you. Not only now to advocate for regular PSA testing, but to advocate for enhanced vigilance should PSA values change while taking finasteride. Finasteride may not increase the true rate of prostate cancer development. But it will obfuscate appropriate interpretation of prostate cancer screening for physicians not informed of both your ongoing prescription and its effect on PSA suppression.
Closing thoughts
Between a finer understanding of PSA velocity and density, accessible imaging tools, and advancements in prostate biopsy and cancer therapeutics, we have made massive inroads towards minimizing the risks of overdiagnosis and overtreatment of prostate cancer.
So we look to the risks of inaction: 36,000 men’s lives in 2026.
We are in the incredibly fortunate position to have the tools we do. Both in early detection and subsequent treatment. We just need to use them—to advocate for them as patients, to understand and utilize them as clinicians—and I believe prostate cancer death could be all-but-eradicated by modern medicine.
For a list of all previous weekly emails, click here.
References
1. Kratzer TB, Mazzitelli N, Star J, Dahut WL, Jemal A, Siegel RL. Prostate cancer statistics, 2025. CA Cancer J Clin. 2025;75(6):485-497. doi:10.3322/caac.70028
2. Wilkinson AN, Ellison LF, Zhang SX, et al. Canadian prostate cancer trends in the context of PSA screening guideline changes. Curr Oncol. 2025;32(12):669. doi:10.3390/curroncol32120669
3. Hugosson J, Carlsson S, Aus G, et al. Mortality results from the Göteborg randomised population-based prostate-cancer screening trial. Lancet Oncol. 2010;11(8):725-732. doi:10.1016/S1470-2045(10)70146-7
4. Alpert PF. New evidence for the benefit of prostate-specific antigen screening: Data from 400,887 kaiser permanente patients. Urology. 2018;118:119-126. doi:10.1016/j.urology.2018.02.049
5. Schröder FH, Hugosson J, Carlsson S, et al. Screening for prostate cancer decreases the risk of developing metastatic disease: findings from the European Randomized Study of Screening for Prostate Cancer (ERSPC). Eur Urol. 2012;62(5):745-752. doi:10.1016/j.eururo.2012.05.068
6. Liss MA, Ehdaie B, Loeb S, et al. An update of the American urological association white paper on the prevention and treatment of the more common complications related to prostate biopsy. J Urol. 2017;198(2):329-334. doi:10.1016/j.juro.2017.01.103
7. Archived: How Did the USPSTF Arrive at This Recommendation?: Prostate Cancer: Screening. May 11, 2012. Accessed February 11, 2026. https://www.uspreventiveservicestaskforce.org/uspstf/document/how-did-the-uspstf-arrive-at-this-recommendation-/prostate-cancer-screening-2012
8. Schoen MW, Montgomery RB, Owens L, Khan S, Sanfilippo KM, Etzioni RB. Survival in patients with DE Novo metastatic prostate cancer. JAMA Netw Open. 2024;7(3):e241970. doi:10.1001/jamanetworkopen.2024.1970
9. Weiner AB, Matulewicz RS, Eggener SE, Schaeffer EM. Increasing incidence of metastatic prostate cancer in the United States (2004-2013). Prostate Cancer Prostatic Dis. 2016;19(4):395-397. doi:10.1038/pcan.2016.30
10. Ornstein DK, Smith DS, Rao GS, Basler JW, Ratliff TL, Catalona WJ. Biological variation of total, free and percent free serum prostate specific antigen levels in screening volunteers. J Urol. 1997;157(6):2179-2182. doi:10.1016/s0022-5347(01)64708-6
11. Tchetgen MB, Song JT, Strawderman M, Jacobsen SJ, Oesterling JE. Ejaculation increases the serum prostate-specific antigen concentration. Urology. 1996;47(4):511-516. doi:10.1016/s0090-4295(99)80486-5
12. Kalish LA, McKinlay JB. Serum prostate-specific antigen levels (PSA) in men without clinical evidence of prostate cancer: age-specific reference ranges for total PSA, free PSA, and percent free PSA. Urology. 1999;54(6):1022-1027. doi:10.1016/s0090-4295(99)00349-0
13. Ng ABCD, Asif A, Agarwal R, et al. Biparametric vs multiparametric MRI for prostate cancer diagnosis: The PRIME diagnostic clinical trial: The PRIME diagnostic clinical trial. JAMA. 2025;334(13):1170-1179. doi:10.1001/jama.2025.13722
14. Hu JC, Assel M, Allaf ME, et al. Transperineal vs transrectal prostate biopsy-the PREVENT randomized clinical trial. JAMA Oncol. 2024;10(11):1590. doi:10.1001/jamaoncol.2024.4000
15. Rezaee ME, Meeks W, Galen E, Helsel A, Pavlovich CP. Examining variation in prostate biopsy practices in the United States: An analysis of American urological association census data. Urology. 2026;208:9-13. doi:10.1016/j.urology.2025.10.039
16. Shoag JE, Mittal S, Hu JC. Reevaluating PSA testing rates in the PLCO trial. N Engl J Med. 2016;374(18):1795-1796. doi:10.1056/NEJMc1515131
17. Tsodikov A, Gulati R, Heijnsdijk EAM, et al. Reconciling the effects of screening on Prostate Cancer mortality in the ERSPC and PLCO trials. Ann Intern Med. 2017;167(7):449-455. doi:10.7326/M16-2586
18. Thompson IM, Goodman PJ, Tangen CM, et al. The influence of finasteride on the development of prostate cancer. N Engl J Med. 2003;349(3):215-224. doi:10.1056/NEJMoa030660
19. Etzioni RD, Howlader N, Shaw PA, et al. Long-term effects of finasteride on prostate specific antigen levels: results from the prostate cancer prevention trial. J Urol. 2005;174(3):877-881. doi:10.1097/01.ju.0000169255.64518.fb
20. Sarkar RR, Parsons JK, Bryant AK, et al. Association of treatment with 5α-Reductase inhibitors with time to diagnosis and mortality in prostate cancer. JAMA Intern Med. 2019;179(6):812-819. doi:10.1001/jamainternmed.2019.0280