In this episode, Peter takes a deep dive into prostate cancer screening, explaining why advanced and metastatic diagnoses continue to rise despite the availability of screening tools, and what can be done to reverse this trend. He breaks down what PSA actually measures and why it is far more informative when tracked over time rather than interpreted as a single value, and he explores how tools like MRI, PSA density, PSA velocity, and improved biopsy techniques can both reduce unnecessary procedures and improve the detection of aggressive cancers. Peter also discusses the role of active surveillance in avoiding overtreatment for low-risk cases, examines the flawed evidence that has historically been used to argue against PSA screening, and highlights how medications like finasteride can suppress PSA levels and potentially mask warning signs if not properly accounted for. Ultimately, he makes a compelling case for the importance of regular PSA testing as a key strategy in the effort to eliminate prostate cancer mortality.
Subscribe on: APPLE PODCASTS | SPOTIFY | RSS | OVERCAST
“My mission for The Peter Attia Drive has always been to provide you with the most rigorous, evidence-informed insights on longevity. To do that without cluttering your experience with ads, we rely entirely on our premium members. If you’d like to support the work that makes this mission possible, consider becoming a premium member.”
– Peter
We discuss:
- The failure of current prostate cancer screening guidelines, and the rise in advanced disease despite available tools [2:30];
- PSA screening fundamentals: benefits, harms, and the guideline shift driven by overdiagnosis concerns [5:30];
- The impact of reduced PSA screening: rising rates of late-stage prostate cancer and worsening population-level outcomes [12:00];
- How modern screening practices use PSA trends, MRI, and new imaging advances to improve accuracy and reduce unnecessary procedures [15:00];
- Advances in prostate biopsy: transperineal approach improves safety and cancer detection [23:00];
- Reducing overtreatment: Gleason scoring and active surveillance in modern prostate cancer care [25:30];
- Reevaluating PSA screening guidelines: how flaws in the PLCO trial undermine the evidence used to argue against PSA screening [29:45];
- Prostate cancer screening today: improved tools, flawed guidelines, and preventable mortality [33:45];
- How finasteride and similar drugs suppress PSA levels and can lead to missed or delayed prostate cancer diagnoses if not properly accounted for [38:00];
- The optimistic future of prostate cancer: modern screening advances and the potential to eliminate mortality [43:15]; and
- More.
Show Notes
*Notes from intro:
- In this special episode, Peter will walk through a single topic in-depth, prostate cancer screening
- This is something Peter is very passionate about ‒ he considers this a really important PSA (no pun intended)
- The full episode and detailed show notes will be available to everyone, regardless of whether or not you’re a premium subscriber
In this episode, Peter discusses
- Why advanced and metastatic prostate cancer diagnoses are rising despite the availability of screening tools
- What PSA actually measures and why it’s more useful when interpreted over time than it is in a single one-off number
- How MRI, PSA density, PSA velocity, and improved biopsy techniques can reduce unnecessary procedures while improving the detection of aggressive cancers
- How active surveillance helps avoid overtreatment in men with low risk cancers
- Why the evidence used to argue against PSA screening was deeply flawed
- How drugs like finasteride (which are commonly used to prevent hair loss) can suppress PSA and lead to missed warning signs if clinicians and patients are not vigilant
- Why Peter thinks regular PSA testing is so important if one of our objectives in life would be to eliminate prostate cancer mortality
The failure of current prostate cancer screening guidelines, and the rise in advanced disease despite available tools [2:30]
- Today Peter wants to dig into something that he feels quite strongly about: prostate cancer screening (specifically PSA testing)
- This is something he thinks we are getting wrong at the population level that is measurably costing lives
- Peter believes the current guidelines have failed men, and he will explain what he thinks a more modern approach to screening should look like in 20226 and beyond
- This is a discussion that is relevant for anybody listening
- As a potential patient
- As someone who cares about a patient
- As a clinician
- At the end of this discussion, Peter will spend some time on something he thinks is dangerously underappreciated: the interaction between a drug called finasteride and how that drug interacts with our ability to interpret PSA correctly
- This is a drug that literally millions of men are taking for hair loss prevention
- Because if you’re on finasteride and your doctor doesn’t understand the implications of that and how that impacts your PSA values, you could be sitting on a ticking time bomb without anybody knowing it
Incidence of prostate cancer in the US
- Prostate cancer is the second leading cause of cancer death in men, with only lung cancer claiming more lives
⇒ About 1 in 8 men will receive a prostate cancer diagnosis at some point in their lives, and this year alone, roughly 36,000 American men will die from it
“What makes this disease both uniquely hopeful and uniquely frustrating is that when you catch it early, the prognosis is outstanding.”‒ Peter Attia
- Early detection of prostate cancer has a 15-year survival rate approaching 97% from first diagnosis
- And we have detection tools that are simple, widely available, and getting more sophisticated every year
- Yet here we are watching the rates of advanced stage prostate cancer climb
The rates at which advanced prostate cancers are showing up is higher today than it was 15 years ago
- Recent data out of both the United States and Canada confirm what many urologists have been warning about for years: we’re catching more and more of these cancers only after they have already metastasized or spread
- That’s what we call stage IV disease [explained in the figure below]
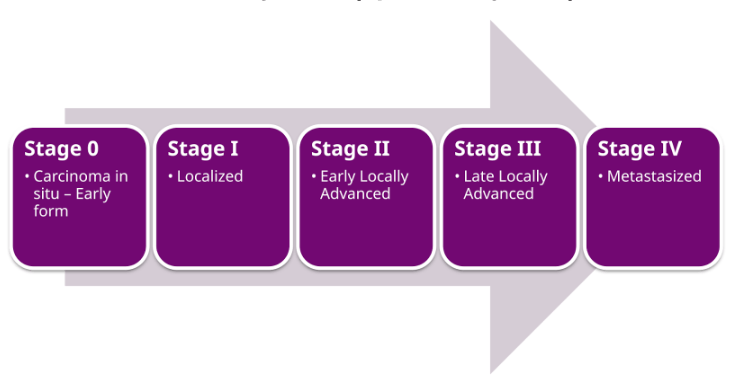
Figure 1. Stage grouping of cancer. Image credit: Wikipedia
⇒ And once you’re at stage IV, the picture changes dramatically: 5-year survival falls to 38%, and the median time from diagnosis to death is about 2.5 years
How did we get here?
- We have the screening tools
- We have effective therapies for early cancers
- Why are outcomes getting worse instead of better?
- And of course, most critically, then what do we need to do about this?
PSA screening fundamentals: benefits, harms, and the guideline shift driven by overdiagnosis concerns [5:30]
To answer any of that, you need to start with the basics of what the PSA actually is because it’s the linchpin of this entire conversation
Peter points out, “Quite frankly, if we had the PSA equivalent for every other cancer, the world could look a heck of a lot different than it does today with respect to cancer mortality.”
- PSA (which stands for prostate-specific antigen), has been available as a blood test since the early ’90s
- It’s a protein made by the prostate gland
- Its biological function is to help keep seminal fluid in a liquid state
- A tiny amount of PSA naturally leaks from the prostate into the bloodstream, and that’s what we measure when we draw a person’s blood
- Here’s the important physiologic point: as the prostate grows (which happens in virtually all men as they age), the amount of PSA in the blood tends to rise proportionally
When the PSA test was first deployed, the logic was simple: if your PSA is unusually high, your prostate is unusually large, and that could mean trouble
- An elevated PSA would prompt an ultrasound
- An abnormal ultrasound would trigger a biopsy
- And cancer on a biopsy would lead to treatment
The early data on this approach were generally reasonable
- Two large trials followed over 14 and 16 years respectively, showed that routine PSA screening cut prostate cancer mortality by 44 and 64% respectively
- That translates to preventing up to 3.1 cases of metastatic disease (which means fatal) for every 1,000 men who went through the screening process
- And the test itself costs nothing, a standard blood draw fully covered by most insurance plans
So you would naturally ask, why wouldn’t we screen everyone?
- Well, here’s where the story takes a turn that Peter thinks had a devastating set of consequences
- Even though the people who made the decision were operating at least somewhat with reasonable logic at the time
The problem was that the straight line from elevated PSA to biopsy was generating too many false alarms
PSA can be elevated for many reasons that have nothing to do with cancer
- Including benign enlargement
- Transient inflammation, such as you might experience during a bout of prostatitis
- Recent sexual activity
- And frankly, even just natural variation
But the biopsies that followed in elevated reading were hardly benign
- The standard approach involved punching a needle through the rectal wall to sample prostatic tissue
- So now you’re creating a contaminated pathway from a bacteria-rich environment into what should be a sterile organ
- And even with things like prophylactic antibiotics and preparation, infection rates from these biopsies ran between 5 and 7%, which of course, when you multiply that across the number of people that were having them, is an enormous amount of morbidity
- And prostate infections, of course, are not a joke, especially if they reach into the bloodstream
- They would frequently land men in the emergency room, they could lead to hospitalizations and take people away from work
On top of that, there was the overtreatment issue
- Back in the ’90s and early 2000s, finding cancer on a biopsy almost invariably led to treatment
- And that surgical treatment carried real consequences
- Erectile dysfunction, urinary incontinence, bowel problems
- Especially if radiation was used
- The psychological weight of a cancer diagnosis alone is enormous
And what we’ve come to understand since then is that a significant fraction of the cancers caught during the early PSA era were actually quite indolent
- They were never going to progress to the point of threatening someone’s life
- These men were being subjected to the full burden of cancer treatment for a disease that if left alone would never have harmed them
- There’s a saying in the urology world that says, “Every man will get prostate cancer, but some will die of it.”
- And the idea here is if you live long enough, you’re always going to find prostate cancer inside some cells of the prostate
- That’s not the jugular question
Peter points out, “The jugular question when you identify prostate cancer is, how many of these cases are cases where if left untreated, the cancer will spread from this organ to a distant organ (namely the bone, which is the most common place to spread)?”
Changes in PSA screening
- In response to all of this, the United States Preventative Service Task Force (or USPSTF) made its move, and in 2008, they stopped recommending PSA screening for men over 70
- By 2012, they extended that recommendation against screening to men of all ages
The data they leaned most heavily on came from a large American trial called the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial (or PLCO)
- The prostate arm of this study randomized men to either undergo PSA screening or not, and it found essentially no difference in prostate cancer mortality between the 2 groups
- The implication was that screening saved at best 1.3 lives per thousand men tested
The USPSTF’s calculus went something like this
- If you’re only saving 1/10th of 1% of men while causing serious complications, the math simply doesn’t add up
- Complications are all the things Peter just mentioned: infections, incontinence, erectile dysfunction, this stress that’s associated with all of these things in a meaningful proportion of the others
The USPSTF concluded that the harms of over-diagnosis and overtreatment exceed the benefits [of PSA screening]
- Now, again, Peter can see how one could arrive at that conclusion
- It’s a little bit of a utilitarian argument and there’s a logic to it at the population level, but that logic only holds if the underlying data are sound (which he’ll discuss)
Peter points out, “It’s been 15 years now. If the USPSTF got it right, we should see at most a marginal uptick in late stage disease (maybe a fraction of a percent).”
- So the obvious next question is, what has actually happened over the past 15 years?
The impact of reduced PSA screening: rising rates of late-stage prostate cancer and worsening population-level outcomes [12:00]
What has actually happened over the past 15 years?
The data are now in and they paint a very troubling picture…
- When you group together all prostate cancers caught at stages I through III, the 5-year survival exceeds 99% (these are non-metastatic cancers)
- But at stage IV (which is metastatic disease, tumors that have spread beyond the prostate), survival plummets to 38% with a median survival of about 30 months from diagnosis
The timing of when you find the cancer is not a minor detail, it’s arguably the determining factor in life and death
What does the recent literature show?
- Data published within the last 6 months from both the American and Canadian registries, which builds on the earlier work from Ted Schaeffer and his colleagues in 2016, is that we are finding more and more prostate cancers only after they’ve become metastatic
- This is exactly what many urologists predicted would happen when we pulled back on screening
But what’s even more alarming is that it’s NOT just older men who are affected
- The Canadian data show a 3.7% year over year increase in stage IV diagnoses among men under 75, between 2010 and 2017 [shown in the figure below]
- And this is happening despite a declining total number of cancers detected in that same age group
So it’s not that more men are getting cancer, it’s that we’re only finding it once it’s too late to cure
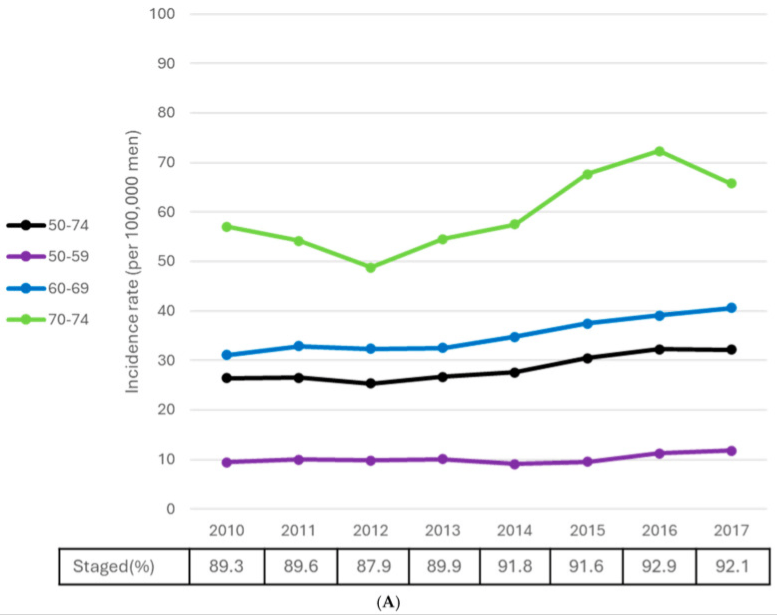
Figure 2. Stage IV prostate cancer age-standardized and age-specific rates, males ages 50-74. Image Credit: Current Oncology 2025
The American data tell a similar story
- Since the 2012 guideline shift, stage II cancer detection has been declining by about 0.1% per year across all ages [see table 3 in CA 2025 and the figure below]
- Meanwhile, stage III has been climbing at 3.3% per year, and stage IV at 6% per year
- Those increases in late stage detection far outpace the modest 0.8% annual increase in total cases
So the interpretation here is not complicated and it is not ambiguous: we stopped looking for early disease, so we started finding it late
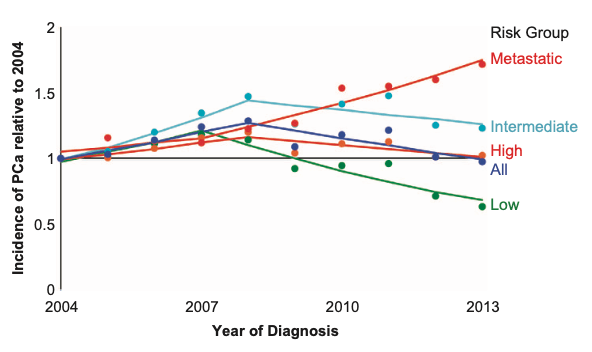
Figure 3. Incidence of prostate cancer by risk group at time of diagnosis. Incidence is normalized to 2004 levels; screening recommendations shifted in 2008 and 2012. Image credit: Prostate Cancer and Prostatic Diseases 2016
⇒ Stage IV cancer is what drives all prostate cancer mortality (or nearly all of it)
- And the trajectory we’re on is the entirely predictable result of a policy that told doctors and patients that looking wasn’t worth the trouble
- Peter thinks the Canadian researchers summarized this paradox well, and he’s paraphrasing: newer treatments have extended survival for men with metastatic disease, but overall survival across the population has actually worsened compared to when screening was recommended because we now have so many more men presenting with incurable cancer
Peter contends, “It doesn’t have to be the story. It just shouldn’t be this way.”
How modern screening practices use PSA trends, MRI, and new imaging advances to improve accuracy and reduce unnecessary procedures [15:00]
- This is where Peter wants to pivot because he thinks this is the most important part of the conversation and the most hopeful
“The USPSTF built its guidance at a time when our tools were far cruder than they are today.”‒ Peter Attia
Initial use of PSA for screening and the treatment that followed
- The old pipeline, as Peter talked about a few minutes ago, was essentially PSA goes up, you get an ultrasound
- Ultrasound looks somewhat concerning, you get a biopsy
- If the biopsy finds any type of cancer, you get treated
- And there was very little nuance in that algorithm
But the way we deploy PSA in 2026, and quite frankly, over the last 5 years, looks poignantly different
Peter points out, “The single biggest conceptual shift is this: PSA is most valued as a longitudinal trend, not a single snapshot.”
The normal range of PSA values
- Any individual PSA reading is inherently noisy
- Day-to-day values can fluctuate by as much as 15%
- If a man has recently ejaculated, his PSA can spike by as much as 40%
- And the person-to-person variability is staggering
- A healthy man in his 60s might have a median PSA of 1.0 to 1.2
- But the 95th percentile for that exact same demographic in men with no prostate cancer extends all the way up to 4.9
- The table below shows the variability between the median PSA and the 95th percentile level of PSA
- It breaks it down by decade of life
The point here is that no single PSA value in isolation tells you very much
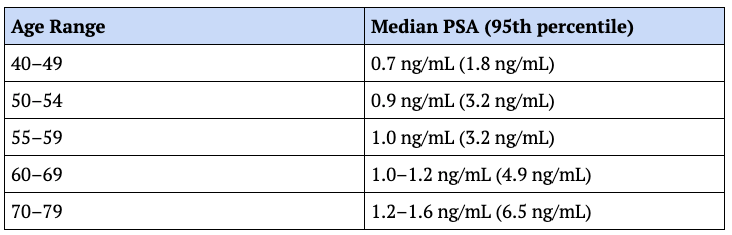
Figure 4. Median PSA by age in men without prostate cancer (95th percentile). Data from: Urology 1999
PSA velocity is superior to a one-time measure of PSA
- What tells you something is the trajectory, and this is what we call PSA velocity: mapping a patient’s baseline over time and tracking the rate of change of that PSA value
- The prostate naturally enlarges with age, so some gradual PSA increase is expected, but rapid increases are cause for suspecting something pathological
- Either the prostate is enlarging abnormally, a higher proportion of PSA is leaking into the blood relative to the amount of prostate, tissue, or both
- And disrupted prostate architecture and vascular leakage are the hallmarks of malignant growth
- In general, if we look at increases sustained over an 18-month window and use different cutoffs depending on the baseline value
- So if we took a patient with a baseline PSA of <4 ng/mL
- And in Peter’s practice, that’s the norm ‒ most men are going to walk around with a baseline PSA <4
⇒ A sustained rise by more than about 0.35 bg/mL/yr over 18 months is generally our red flag
- In patients with a PSA value that starts >4, we would let that increase to about a 0.75 ng/mL/yr bump before we would flag that
A tangible example
- A 55-year-old patient with a PSA of 1.3
- So that’s slightly above the median for his age, but Peter is not alarmed, especially because he has longitudinal data on him and he realizes that this is about where he settles out
- So that one time he was 1.6, Peter didn’t overreact because 2 months later or 3 months later, he was back to 1.3
- Now, if he comes back a year later at 1.7, and then 6 months after that comes back at 1.9
- That’s an increase of 0.6 over an 18-month period of time, working out to a velocity of 0.4 per year
- Now Peter is paying attention
That sustained acceleration is the signal Peter is looking for, and it’s personalized to him
“This is a great example of precision medicine, and incredibly, it costs essentially nothing beyond showing up for a routine blood draw.”‒ Peter Attia
- Again, the blood test itself costs dollars
- But even with his velocity flagging a concern, we no longer would just jump to a biopsy here
The next step would now be an MRI, which serves as a much higher resolution test, and it’s much less invasive than proceeding to a biopsy
The MRI gives us yet another metric, which is PSA density
- PSA density is simply the PSA concentration in the blood divided by the volume of the prostate (provided by imaging)
- You could do that with an ultrasound
- But with the MRI, you’re getting 2 for 1 because you’re getting a better imaging study and of course you’re getting volume
Here’s the logic
- A healthy prostate grows, PSA and volume tend to increase in approximate proportionality
- But when you have a malignancy, the tumor can disrupt the normal architecture of the prostate that normally contains PSA to the gland or alters the endothelia running through the prostate
- And in this situation, PSA starts escaping into the bloodstream at a rate that outpaces what you’d expect from the size of the gland alone
So if PSA is rising disproportionately to prostate volume, that’s a strong indicator that something is wrong, and it gives us that signal before anybody needs to think about a needle
Better screening is available now
Peter makes the point, “When you put your PSA velocity from serial blood tests together with PSA density from MRI, the discriminating power of PSA screening today is dramatically better than what existed when the USPSTF made its call.”
Now, if we focus in on MRI for a moment
- There’s been a really significant development that Peter thinks is going to meaningfully change the accessibility equation
- Beyond PSA density, the main value of MRI in this algorithm is that it can identify suspicious lesions or regions in the prostate without invasive probing
- It can credibly rule in or rule-out the need for a biopsy
- And if a biopsy is warranted, it guides the needle to the areas most likely to hand us a diagnosis
- The obstacle historically was that the gold standard prostate MRI, called a multiparametric MRI, requires a contrast called gadolinium
- And that meant higher costs, the need for a medical professional to administer an IV injection, and significantly more time in the scanner
- All of this limited patient throughput and made pre-biopsy MRI impractical in many scenarios
But a trial in 2025 called the PRIME trial changed this
- The study directly compared the full multiparametric protocol against a stripped-down contrast-free biparametric version that drops the contrast component altogether
- The multiparametric protocol is a T2-weighted image, a diffusion-weighted image, plus dynamic contrast enhanced imaging
- The goal was to establish non-inferiority
- In other words, the question asked was, “Does this stripped-down version perform at least as well as the gold standard?”
And the results were essentially a dead heat [the data is shown in the figure below]
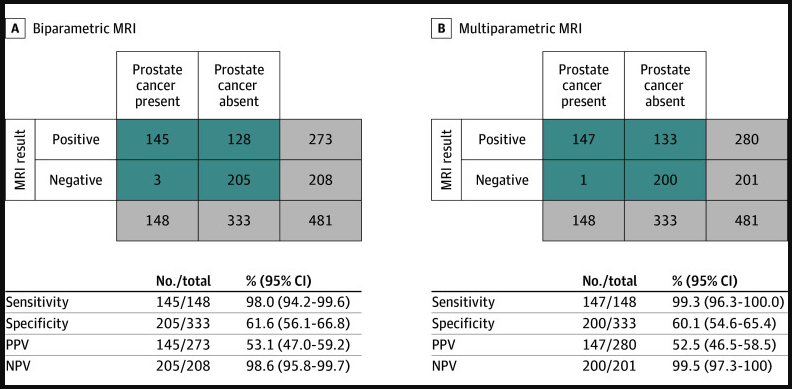
Figure 5. Test performance characteristics of biparametric and multiparametric MRI. Image credit: JAMA 2025
- The biparametric MRI identified clinically significant cancer in 143 out of 490 men who had been flagged by PSA screening
- Whereas the full multiparametric MRI complete with gadolinium caught 145 out of that same group of 490 study participants
- The difference between those detection rates was 0.4 percentage points, which was statistically indistinguishable
And the practical implications here are huge
- Without contrast, the scan takes half the time: 15-20 minutes instead of 30-40
- You don’t need a medical practitioner that’s standing there giving you the contrast and overseeing you
- And of course the costs come down, speed goes up, etc.
Advances in prostate biopsy: transperineal approach improves safety and cancer detection [23:00]
- Now, even with all of these improvements in non-invasive screening, some men are still going to need a biopsy
- If the MRI flags something suspicious and you need tissue to know what you’re dealing with, you’ve only got one place to get it
- So it’s worth talking about the progress that’s been made there as well because the biopsy itself has gotten both safer and more accurate
The traditional approach is a transrectal approach
- A transrectal biopsy [shown in the figure below] carries inherent risk of poking bacteria from the rectum into a sterile space
- It also has its anatomical limitations
- It biases your sampling towards the back of the prostate near the rectal wall, and this can miss the front and base of the gland
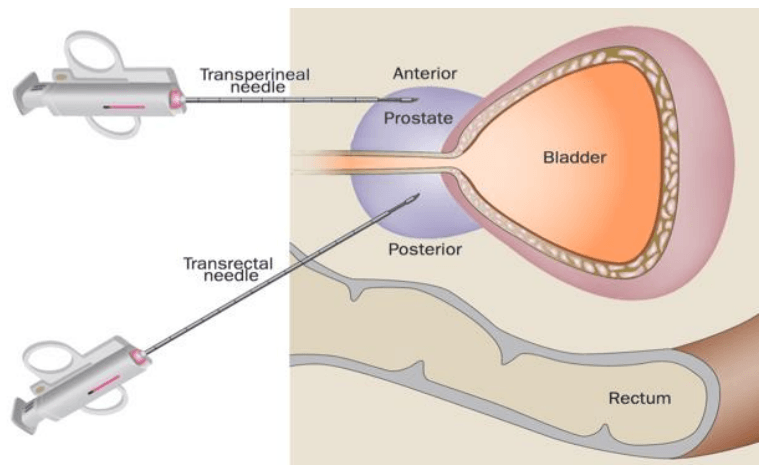
Figure 6. Biopsy approach used for a transperineal versus a transrectal biopsy. Image credit: JAMA Oncology 2024, supplement 1
The alternative to this is called a transperineal approach
- A transperineal biopsy enters through the skin between the anus and the scrotum, and this completely bypasses the rectum
- Which dramatically reduces infection risk
- And it opens up much better access to the anterior and apical portions of the prostate than the transrectal route does (which it tends to miss)
- Ted Schaeffer has been a guest on this podcast many times and is a very close friend of Peter’s ‒ he is also the urologist that works with many of Peter’s patients when they are diagnosed with prostate cancer
- He was involved in a large multicenter phase III trial with over 1,700 patients that compared cancer detection rates and complication profiles between the 2 approaches head to head [the PREVENT trial ]
The transperineal biopsy detected more high grade cancers and fewer low grade ones, which tells you it’s doing a better job of finding tumors that actually matter, generating fewer false negatives from indolent disease and obviously creating less emotional distress
On the safety front
- Not a single patient who underwent the transperineal biopsy in that study developed an infection (zero)
⇒ Compare that to the 5-7% infection rate widely reported in the transrectal approach
- Importantly, the transperineal method doesn’t require prophylactic antibiotics, which is a meaningful benefit both for the patient and for the antibiotic stewardship more broadly as we face an era of antibiotic resistance
Now, adoption isn’t universal yet
- Only about 37% of American urologists currently perform transperineal biopsies versus essentially all of them doing transrectal or being able to
- So there’s a gap between what the evidence supports and what’s happening in practice, but the trajectory is encouraging
Reducing overtreatment: Gleason scoring and active surveillance in modern prostate cancer care [25:30]
- Peter has been building the case for more screening, for more MRI, and for better biopsies
- But he hasn’t forgotten the other side of the original argument, which is the overtreatment problem
- He really takes this seriously because with all of the advances in surgical care, this is NOT a benign operation
- Not every prostate cancer is going to kill you
- Our treatments are effective, but they carry side effects that nobody wants to experience if they don’t have to
Here’s the critical point that Peter thinks changes the overtreatment calculus: we no longer treat every cancer we find
- The decision tree has gotten much more sophisticated
⇒ Ted Schaeffer explains Gleason scoring in episodes #39 and #273
Peter’s quick summary of Gleason scoring
- When a pathologist examines a biopsy specimen, they look at the cells under the microscope and they decide how abnormal the cells look relative to healthy cells, and they use a grading scale called a Gleason score
- You’re getting 2 numbers in that score because they’re looking in 2 places
- So you might get a Gleason 3+3 or 3+4 or 4+4
- And the highest number there is 5
The higher the number, the greater the malignant potential of the cell
- These numbers get compressed into a grade group, which is a scale that is graded 1 through 5
- And again, it’s similarly based on the abnormality of these cells
A grade 1 or 2 is a relatively low risk tumor
- We know the natural history of grade 1 and grade 2 (which are the lower Gleason scores), they behave very differently, and these are cancers that may never spread
- Now, we don’t know that for a fact, but we know that if they’re going to spread, they’re going to do so by progressing into a higher grade Gleason score
- And therefore, for these patients, we can continue to surveil them or do other advanced forms of monitoring (which Peter will talk about soon)
Conversely, if a patient presents with a higher grade group
- Like a grade 3, which would be a Gleason 4+3
- A grade 4, a Gleason 4+4
- Or a grade 5
These are very aggressive tumors and treatment is immediately warranted
Back to grade 1 and grade 2 tumors
- These are patients that have prostate cancer, but our belief is that it is not an imminent threat to their life because it is not clear if these cancers will spread
- And if they’re going to turn into spreading cancers, they’re going to do so by progressing
- In this case, we would look at other blood markers like tests called the phi or the 4Kscore®
- These are things that we talked about in episode #273 (after [2:00:00])
The point is we now turn into an area called active surveillance
- And active surveillance is basically something we’re doing to ensure that treatment will be timely once it is required, but no sooner
The scenario that drove so much of the overtreatment concern 15 years ago
- When you find any cancer, you treat it immediately, and you deal with the consequences
Peter explains, “That’s largely been retired from the urologist playbook.”
How the field has advanced
- The tools and clinical judgments have evolved today to the point where urologists can meaningfully distinguish between dangerous tumors and ones that can be safely watched
- And this is what basically gives us this luxury
And that means that the risk benefit calculation for screening has shifted substantially in favor of screening
Reevaluating PSA screening guidelines: how flaws in the PLCO trial undermine the evidence used to argue against PSA screening [29:45]
- Peter just spent a good amount of time walking through the modern screening toolkit
- And he’s tried to be fair to the original USPSTF reasoning along the way
- He thinks the overtreatment concern was legitimate and the burden of transrectal biopsies was real
- Again, he takes those objections seriously, but there’s something he’s held back on until now
“It’s a failure in the data that I think is so significant that it essentially undermines the entire foundation of the USPSTF position.”‒ Peter Attia
- Urologists have been pointing this out for years, and Peter thinks it’s borderline unbelievable that it hasn’t led to a formal revision of the guidelines
Peter is talking about the PLCO trial
- That’s the study that the USPSTF weighted most heavily when it decided that PSA screening wasn’t worth doing
- This study took a large group of men, randomize half to get regular PSA tests, the other half not to get tested
- Follow them over a decade and compare prostate cancer deaths between the 2 groups
- The study found essentially no difference supporting the conclusion that screening doesn’t meaningfully reduce mortality
But here’s what went wrong
- Peter genuinely had trouble believing this the first time he encountered it
- The control arm (this is the group that was not supposed to be screened) was getting screened anyway
- The investigators may not have been providing the tests, but when they surveyed the participants, somewhere between 40-60% of men in the no screening group reported having had a PSA test within the year
- And this wasn’t a one-time contamination event ‒ that pattern held every single year of the study
- So by the final survey, over 90% of the men in the control arm had undergone at least 1 PSA test
- In some years, more men in the supposed control arm reported screening than in the intervention arm
- Below is the figure from the 2016 letter to the editor in the New England Journal of Medicine
- You can see this for yourself because it is quite striking
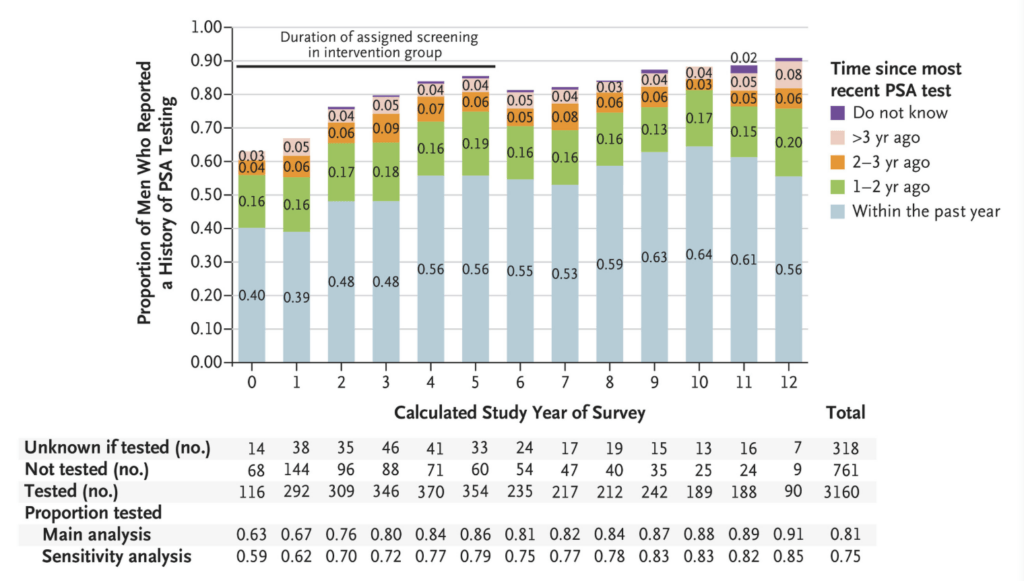
Figure 7. Survey of respondents in the “no PSA testing” control arm of the PLCO study, reporting time since their most recent PSA test. Image credit: NEJM 2016
- The bottom line is, this was a study comparing PSA screening to [no] PSA screening and then concluding that screening doesn’t work when they didn’t find a difference
- That is the bedrock upon which the USPSTF built a recommendation that has affected tens of millions of men
⇒ Now, this data contamination wasn’t published widely until 2016, well after the original guideline change
Once it was out there, a group of researchers went back to the PLCO data in 2017 and did something really clever
- They mathematically accounted for the frequency of screening within the contaminated control arm, calculated a mean and lead time (essentially how much earlier cancers were caught in men who were screened more regularly), and then imputed a mortality reduction based on earlier diagnoses
- When you run the analysis that way, correcting for the contamination that made the original study essentially uninterpretable, the same PLCO data show a 27-32% reduction in prostate cancer mortality attributable to regular PSA testing
- Peter just wants to sit with that for a second
The same data set, the same patients analyzed honestly, and it shows that screening prevents roughly a quarter to a third of prostate cancer deaths, and yet the USPSTF has not updated its guidelines since 2018
- It still does not recommend PSA testing for men 55-69 and still recommends against it for men over 70
“I think it’s long past time for the USPSTF to revisit this.”‒ Peter Attia
- The same is true of organizations like the Canadian Task Force on Preventive Healthcare [guidelines] and the American Association of Family Physicians [guidelines]
Peter emphasizes, “Until they do so, I think we have reason to be confident in taking this into our own hands.”
Prostate cancer screening today: improved tools, flawed guidelines, and preventable mortality [33:45]
A synthesis of everything covered so far
- Peter realizes it’s been a lot
- We started with the observation that pulling back on PSA screening led to a measurable ongoing increase in the incidence of incurable prostate cancers
- That’s not speculation ‒ it’s in the data; it’s black and white from multiple countries
We then walk through how PSA testing has evolved
- A single PSA value taken once every few years was never a perfect screening tool
- It’s too noisy, too much individual variation
Peter acknowledges, “That part of the old critique was fair.”
But PSA velocity, PSA density, contrast-free MRI, and transperineal biopsies along with active surveillance for low grade disease taken together represent a fundamentally different screening paradigm than what existed when the USPSTF issued its recommendations
“The ability to catch dangerous (lethal cancers) early while avoiding the harms of overdiagnosis and overtreatment has improved by an order of magnitude.”‒ Peter Attia
And then we looked at the data that supposedly justified the shift away from screening 15 years ago
- We found that the cornerstone study was so contaminated as to be essentially meaningless
A proper analysis of the same data clearly supports screening very strongly
So where does that leave us?
- Well, prostate cancer is the #2 cancer killer of men and it’s not receiving the urgency it deserves
- Peter goes further, “I think it’s unconscionable that we are still losing men to a disease that in many cases could have been caught years before it became lethal.”
- This is something he wants people to really internalize because it goes to the biology of the disease itself
- Peter has talked about this many times on previous podcasts, but he doesn’t think he can talk about it enough
Cancer falls into roughly 2 categories
- 1 – There are those that progress in a relatively predictable, stepwise fashion moving through detectable precursor stages before becoming truly dangerous
- 2 – And then there are those that don’t
- They can emerge suddenly, metastasize quickly, and evade early detection no matter how aggressively you screen
- Pancreatic cancer is the classic example of the latter
- And sadly, many breast cancers also fall into that category
- But prostate cancer, along with colorectal cancer, fit squarely in the first group
Prostate cancer tends to follow a very predictable path from early localized disease to advanced metastatic disease
- And that biological reality is precisely what makes a screening detectable tool (like the PSA) so powerful
- And what makes any deaths, let alone the 36,000 a year in this country alone from prostate cancer, so completely tragic
Peter’s point is, “This isn’t a cruelty of biology, it’s a failure of our implementation.”
Peter has made this comparison before, but he thinks it’s worth repeating
- Imagine we had a breast cancer detection tool that cost less than a cup of coffee
- It can be incorporated into a routine blood test, and was capable of flagging cancer years before it spread
- In what universe would we not deploy that tool as widely and aggressively as humanly possible?
For prostate cancer, that tool does exist
- Your primary care doctor can add it to your next blood draw (it might already be there)
The barrier isn’t technology, it’s awareness, it’s education, and most of all, it’s outdated guidelines
- In Peter’s practice, he’s seen this play out countless times.
- Patients will come with PSA data from other providers (sometimes years of it), and when we calculate their velocity, the red flags are right there
- We send them for an MRI, depending on what it shows, a biopsy
- And we’ve caught several cancers right at that stage where had we waited another year or two, they likely would’ve been metastatic
- They were Gleason 3+4 or 4+3 that were right in that sweet spot for treatment
How finasteride and similar drugs suppress PSA levels and can lead to missed or delayed prostate cancer diagnoses if not properly accounted for [38:00]
- Before closing, Peter wants to spend some time on a topic related to this, but a little bit oblique to it
- It’s becoming increasingly prevalent and it’s something we need to be alarmed about ‒ physicians and patients alike need to be aware of this
The effect of a class of drugs called 5α-reductase inhibitors on prostate cancer screening
- The most common 5α-reductase inhibitors are finasteride and dutasteride
Peter points out, “The use of finasteride has exploded in recent years, largely driven by men using it to prevent or treat hair loss.”
⇒ Check out further discussion of finasteride in the context of hair loss [episode #43], testosterone use [episode #180], and risk for prostate cancer development [episode #273]
- As far as we can tell from the data, finasteride does NOT cause prostate cancer
- But what it absolutely unequivocally does is suppress PSA in a way that can completely mask the presence of cancer if the treating physician doesn’t know how to adjust for it
Here’s the mechanism
- Finasteride blocks the conversion of testosterone into dihydrotestosterone (or DHT) in the prostate
- And that reduction in local DHT also dramatically reduces PSA production
⇒ As a practical matter, you can expect PSA to drop by about a third within 6 months of starting finasteride and to be roughly cut in half by a year
- That suppressed baseline persists for as long as the patient stays on the drug
Peter emphasizes, “Let me state this as clearly as I can. If a man has been on finasteride for a year or longer, his PSA value needs to be at a minimum doubled to be interpreted correctly.”
The correction factor Peter uses increases with duration of use
⇒ In his practice, he multiplies [the PSA measurement] by 2.3 for patients who have used the drug for 2-7 years and by 2.5 for patients who have used the drug for 7 years or longer
- While the FDA label recommends multiplying by 2, Peter does this based on data from the Prostate Cancer Prevention Trial performed in the late 90s and early 2000s
- As well as a follow-up analysis from 2005 performed by Ruth Etzioni and her colleagues [this study showed how to adjust the effect of finasteride on PSA measurement based on how long a person has been taking the drug, summarized in the table below]
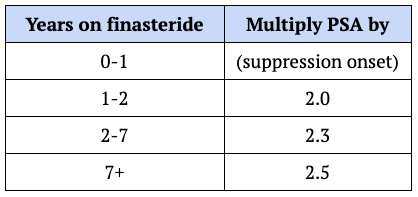
Figure 8. PSA adjustment factor based on duration of finasteride use. Based on: Journal of Urology 2005
⇒ For PSA velocity, the practical implication is simpler, but equally important: after about 12 months on finasteride, you should NOT expect your PSA to rise at all
- The ongoing suppression from the drug should roughly offset the natural age-related increase in PSA
- In fact, in a large study of 19,000 older men, those on finasteride who were cancer-free actually showed an average 2% annual decrease in PSA
So any upward movement [of PSA velocity] in a man on long-term finasteride should be treated as a serious warning sign
Here’s where Peter gets concerned
- Most general practitioners are NOT aware of this, it appears
- A retrospective analysis of VA patients undergoing PSA screening found that men on finasteride were diagnosed with metastatic cancer at a rate more than double that of men not taking 5-alpha reductase inhibitors (6.7% versus 2.9%)
The doctors were reading the raw PSA numbers at face value, not realizing they needed to double them or more [for men taking finasteride]
- The 2 groups had nearly identical PSA levels at the time of cancer diagnosis, about 6.8 versus 6.4
- But once you correct for the finasteride induced suppression, the true comparison is more like 13.5 versus 6.4
- Those are wildly different clinical pictures and the physicians unfortunately missed it
“This is in some ways an even more tragic version of the same problem Peter has been talking about for the past hour.”‒ Peter Attia
- Because even in men who were getting screened, the screening was rendered useless because the physician didn’t understand how to correct the results
Peter emphasizes the clinical relevance, “That’s especially urgent because many men are getting finasteride prescriptions from online clinics and then not even mentioning it to their primary care doctors because they don’t think it matters.”
- So even a physician who does understand the PSA interaction can’t account for it if they don’t know the patient’s taking it
Takeaway for the listener ‒ if you’re listening to this and you’re taking finasteride, the onus may fall on you
- Please be sure that you not only advocate for regular PSA testing, but make sure that your doctors know about every prescription, including, and especially those that involve 5α-reductase inhibitors, and ensure that whoever is interpreting your PSA understands both the drug and its suppressive effects
Again, finasteride does not appear to increase your actual risk of developing prostate cancer, but it can absolutely prevent your doctors from catching it on time
The optimistic future of prostate cancer: modern screening advances and the potential to eliminate mortality [43:15]
The evidence points to a place of cautious but genuine optimism
- Between our understanding of PSA velocity and density, the increasing accessibility of very high quality MRI, the advent of transperineal biopsy, and the sophistication of active surveillance protocols for low grade disease (such as those Gleason 3+3s), the field has made enormous progress in minimizing the very real risks of overdiagnosis and overtreatment that motivated the USPSTF’s original positioning
- And then against that, we have to weigh the cost of doing nothing: which is a little over 36,000 lives this year in the United States
We are in an extraordinarily fortunate position
- We have the tools for early detection that are cheap, simple, and effective
- We have treatments that work
- We have the ability right now to catch most prostate cancers before they become fatal
Peter makes the point, “We just need to use what we have, to advocate for it as patients, and to understand it as clinicians. And I genuinely believe that if we do, prostate cancer deaths can be all but eradicated.”
- Prostate cancer doesn’t have to be the second leading cause of cancer death in men
- It could not even be in the top 20
- A summary of how Peter approaches PSA testing in his practice can be found in a recent newsletter published on 4/19/26
- You can also find all of the studies and references from this episode in the “selected links” section below
Selected Links / Related Material
Prostate cancer statistics for the US: [4:45, 12:15]
- Prostate cancer statistics, 2025 | AC: A Cancer Journal for Clinicians (Kratzer et al. 2025)
- Increasing incidence of metastatic prostate cancer in the United States (2004-2013) | Prostate Cancer and Prostatic Diseases (A Weiner et al. 2016)
Prostate cancer statistics for Canada: Canadian Prostate Cancer Trends in the Context of PSA Screening Guideline Changes | Current Oncology (A Wilkinson et al. 2025) | [4:45, 12:15]
USPSTF rational for 2012 guidelines on prostate cancer screening: How did the USPSTF arrive at this recommendation, prostate cancer: screening | US Preventive Services Task Force (2012) | [11:00]
USPSTF prostate cancer screening recommendation: Final recommendation statement, prostate cancer: screening | US Preventive Services Task Force (2018) | [11:00, 33:15]
Overall survival of stage IV prostate cancer: Survival in Patients With De Novo Metastatic Prostate Cancer | JAMA Network Open (M Schoen et al. 2024) | [12:15]
USPSTF cancer screening recommendations: How Did the USPSTF Arrive at This Recommendation? Prostate Cancer: Screening | US Preventive Services Task Force (2012) | [11:00, 15:15, 20:15]
Biparametric MRI can be used to identify prostate cancer, the PRIME trial: Biparametric vs multiparametric MRI for prostate cancer diagnosis: The PRIME diagnostic clinical trial | JAMA (A Ng et al. 2025) | [21:30]
Use of a transperineal biopsy improves patient outcomes, the PREVENT trial: Transperineal vs Transrectal Prostate Biopsy—The PREVENT Randomized Clinical Trial | JAMA Oncology (J Hu et al. 2024) | [24:00]
Ted Schaeffer explains Gleason scoring of prostate cancer on The Drive: [26:15]
- #39 – Ted Schaeffer, M.D., Ph.D.: How to catch, treat, and survive prostate cancer (February 4, 2019)
- #273 ‒ Prostate health: common problems, cancer prevention, screening, treatment, and more | Ted Schaeffer, M.D., Ph.D. (October 2, 2023)
Advanced prostate cancer blood biomarker tests: [28:45]
- Prostate Health Index (phi) | Beckman Coulter (2023)
- The 4Kscore® Test | 4Kscore (2022)
PLCO trial: Mortality results from a randomized prostate-cancer screening trial | NEJM (G Andriole et al. 2009) | [30:30]
Letter to the editor about PSA testing in the control arm of the PLCO trial: Reevaluating PSA Testing Rates in the PLCO Trial | NEJM (J Shoag, S Mittal, J Hu 2016) | [31:45]
Reanalysis of data from the PLCO trial: Reconciling the Effects of Screening on Prostate Cancer Mortality in the ERSPC and PLCO Trials | Annals of Internal Medicine (A Tsodikov et al. 2017) | [32:15]
Canadian Task Force on Preventive Health Care guidelines for prostate cancer screening: Prostate cancer | Canadian Task Force on Preventive Health Care (2014) | [33:30]
American Association of Family Physicians guidelines for prostate cancer screening: Screening for Prostate Cancer: Recommendation Statement | American Association of Family Physicians (2018) | [33:30]
Peter discusses finasteride in the context of hair loss and testosterone use and also the risk for prostate cancer development: [38:45]
- #39 – Ted Schaeffer, M.D., Ph.D.: How to catch, treat, and survive prostate cancer (February 4, 2019)
- #43 – Alan Bauman, M.D.: The science of male and female hair restoration – how to protect, enhance, and restore the appearance and health of the hair and scalp (March 4, 2019)
- #180 – AMA #28: All things testosterone and testosterone replacement therapy (February 2, 2024)
- #260 ‒ Men’s Sexual Health: why it matters, what can go wrong, and how to fix it | Mohit Khera, M.D., M.B.A., M.P.H. (June 26, 2023)
- #273 ‒ Prostate health: common problems, cancer prevention, screening, treatment, and more | Ted Schaeffer, M.D., Ph.D. (October 2, 2023)
- #274 – Performance-enhancing drugs and hormones: risks, rewards, and broader implications for the public | Derek: More Plates, More Dates (October 9, 2023)
- #316 – AMA #63: A guide for hair loss: causes, treatments, transplants, and sex-specific considerations (September 9, 2024)
Multiply PSA values by 2 (or more) for men taking finasteride, the Prostate Cancer Prevention Trial: [40:15]
- The influence of finasteride on the development of prostate cancer | NEJM (I Thompson et al. 2003)
- Long-term effects of finasteride on prostate specific antigen levels: results from the prostate cancer prevention trial | Journal of Urology (R Etzioni et al. 2005)
Retrospective analysis of VA patients found men on finasteride were diagnosed with metastatic cancer 2x more often than men not taking this drug: Association of Treatment With 5α-Reductase Inhibitors With Time to Diagnosis and Mortality in Prostate Cancer | JAMA Internal Medicine (R Sarkar et al. 2019) | [41:15]
Newsletter on prostate cancer screening: We have the tools to catch prostate cancer early—why aren’t we using them? | PeterAttiaMD.com (N Nelson et al. 2026) | [44:30]
People Mentioned
Ted Schaeffer (Chair of the Department of Urology at Feinberg School of Medicine, Program Director of the Genitourinary Oncology Program at Northwestern University; he specializes in prostate cancer) [13:00, 24:00]