If you’ve been following us for a while, you are no stranger to LDL cholesterol (LDL-C). You’re also no stranger to its most harrowing consequence—atherosclerotic cardiovascular disease (ASCVD), responsible for over 400,000 deaths in the USA each year. And you’re familiar with the causal factor in atherosclerosis development: ApoB-containing particles (the vast majority of which are LDLs), which convey risk by entering arterial intimal layers and setting off the chain reaction that leads to atherosclerotic plaques.
You know our stance: the fact that ASCVD is the leading killer of adult Americans is a tragedy, especially given how good our tools are for managing risk. Statins are widely available, inexpensive, and effective tools for pharmacologic reduction of apoB (and its lipid surrogate measures, non-HDL-C and LDL-C) that are well-tolerated by the great majority of patients. When those tools aren’t enough, or in circumstances where statins aren’t tolerated, we have increasingly accessible tools like PCSK9 inhibitors, ezetimibe, and bempedoic acid to enhance or replace statin therapy. And we know that ASCVD risk develops over decades of elevated exposure—so if we intervene as early and aggressively as possible, the effective risk reduction from lipid lowering therapy can compound throughout a person’s life.
Released in March, new guidelines1 endorsed by nearly a dozen health organizations—including the American Heart Association (AHA) and American College of Cardiology (ACC)—have meaningfully moved the needle towards earlier intervention for prevention of ASCVD.
But has the needle moved far enough?
Earlier is better
The most striking shift in the guidelines is in who they apply to: for people as young as 30 years old, lipid-lowering therapy is now recommended in patients with LDL-C above 160 mg/dL, starting with a moderate intensity statin. An updated risk calculator2 is now available, tuned to calculate risk in patients 30-79 years old where the previous edition only went as young as 40. And it’s now able to calculate the risk of developing ASCVD or heart failure further into the future: not just in the next ten years, when almost no 30–40 year old will be at risk, but into the next 30 years as well.
Additional screening for cardiovascular risk is now also recommended. In addition to genetic screening for familial hypercholesterolemia (FH), the new guidelines recommend all patients undergo at least one blood test for Lp(a) concentration—a potentially severe risk factor for ASCVD, impacting approximately 20% of people. Coronary artery calcium scans (CAC)—a common imaging assessment of atherosclerosis in the three arteries supplying the heart (which I’ve written about and explored on the podcast)—is recommended to inform treatment of patients in borderline and intermediate risk categories. The recommendations suggest apoB testing may be “reasonable” when stratifying risk, or in assessing patients after they’ve reached their LDL-C goals.
Critically, the guidelines recommend more aggressive management by lipid lowering therapy, and now utilize specific numerical cutoffs instead of percentage-based reductions that could vary dramatically from patient to patient. For very high risk patients, LDL-C targets are now below 55 mg/dL (the 2nd percentile for LDL-C, according to the NHANES). For high risk, below 70 mg/dL (10th percentile). And for borderline/intermediate risk, below 100 mg/dL (40th percentile). They also now recommend using CAC scores in excess of 1000 as a criterion to aggressively reduce LDL-C below 55 mg/dL.
Altogether, this is undoubtedly a victory for ASCVD prevention. It may be the single biggest leap forward in the field from the standpoint of recommendations, and we applaud this very enthusiastically. Earlier and more aggressive treatment recommendations will, over decades, save millions of people from early death as a result of atherosclerosis.
Still, I will never tire of sounding like a broken record on this topic, because it is the largest public health problem of our time. We are all at risk of ASCVD—if we live long enough to get it. Therefore, we cannot be aggressive enough in addressing this problem head on.
Our research team spends hundreds of hours each month vetting studies and distilling dense literature to deliver evidence-informed insights on health and longevity. If you find value in our work, consider becoming a premium member and supporting our mission.











Even earlier is even better
The development of ASCVD accumulates throughout life. Risk is expressed as an area under the curve, with LDL-C being on the Y axis, and time on the X axis (see below). The higher your LDL-C is, the more risk you accumulate. The longer you are alive, the more risk you accumulate. How high your LDL-C is and for how long is the key calculation for atherosclerosis risk.
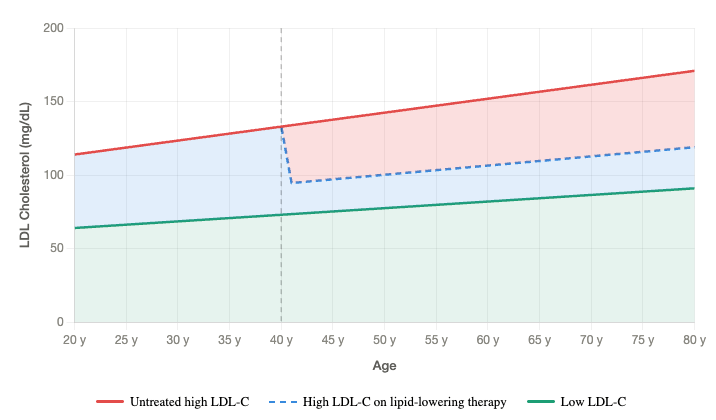
This is why intervening earlier is better: if you cut your LDL-C by 40 mg/dL in your 30s, by your 50s, you have 20 years of 40 mg/dL reduction under your belt. You have decades of accumulating risk reduction.
Clinical trials help us paint this picture—meta-analyses find that, for each 38 mg/dL reduction in LDL-C, risk of major cardiovascular events drops by 12% after one year. But it drops by 20% after 3 years, and by 29% after 7. The reduction in risk compounds through time.3
If we move to Mendelian Randomization (MR), by following people with genetic variants that naturally reduce LDL-C levels, we can study how risk of ASCVD is impacted by lifelong variation rather than pharmacological intervention. If risk reductions compound through time, we’d expect even greater reduction here: clinical trials typically intervene in the 40s, 50s, or later; genetic variation acts throughout a person’s entire life. And this is precisely what is observed—gene variants that lead to reduced levels of circulating LDL-C yield a 54% reduction in ASCVD risk4 for every 38 mg/dL reduction.
The longer a person lives with lower LDL-C, and the lower we get their LDL-C, the greater their protection against ASCVD.
Shifting guidelines to include people in their 30s is progress. That they still only recommend initiating therapy at 160 mg/dL LDL-C, which is the 90th percentile (highest 10%) in the general population, means we are letting significant risk accumulate before acting.
Better targets (but not the right target)
We also know that LDL-C is only a surrogate for the best predictor of risk in atherosclerosis development. That title belongs to apoB particles, which carry LDL-C (as well as VLDL-C and chylomicron remnants) in the blood. The total amount of cholesterol within LDLs isn’t the true driver; how many particles are carrying that cholesterol is. And yet guidelines revolve around LDL-C.
Couching recommendations for apoB testing as something that “may be reasonable” to stratify risk undersells its importance—it is far more reasonable to suggest it is the most important factor for stratifying risk. Dancing around apoB measurement, or holding off until patients have already initiated treatment, risks missing patients that would be taking lipid lowering therapies years or decades earlier—if we only used the definitive test for risk. If we rely on a standard lipid panel, lacking critical measurements like apoB and Lp(a), we turn a blind eye to mountains of evidence that apparently “healthy” people can be at severe risk from atherosclerosis-mediated death.
Progress, not perfection
Earlier initiation of lipid-modulating therapies (including combination therapies), lower LDL-C targets for those at risk, and enhanced screening recommendations are all steps in the right direction. Millions of lives could be saved from early death due to ASCVD.
We have fantastic tools available: statins are inexpensive, side effects real but rare, and multiple statins can be attempted if side effects are present. Even if multiple statins fail, numerous additional tools like ezetimibe, bempedoic acid, and PCSK9 inhibitors are available. And if no single approach is sufficient on its own, these tools can be combined for greater lipid modulation. ASCVD is a solvable problem.
These updated guidelines are good news. But I want to be clear: they’ve left a ton of attainable prevention on the table. Endorsing even earlier and more aggressive lipid lowering interventions would and will save more lives.
For a list of all previous weekly emails, click here.
References
1. Blumenthal RS, Morris PB, Gaudino M, et al. 2026 ACC/AHA/AACVPR/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of Dyslipidemia: A report of the American college of cardiology/American heart association joint committee on clinical practice guidelines. J Am Coll Cardiol. Published online March 13, 2026. doi:10.1016/j.jacc.2025.11.016
2. The American Heart Association PREVENTTM Online Calculator. professional.heart.org. Accessed April 3, 2026. https://professional.heart.org/en/guidelines-and-statements/prevent-calculator
3. Wang N, Woodward M, Huffman MD, Rodgers A. Compounding benefits of cholesterol-lowering therapy for the reduction of major cardiovascular events: Systematic review and meta-analysis. Circ Cardiovasc Qual Outcomes. 2022;15(6):e008552. doi:10.1161/CIRCOUTCOMES.121.008552
4. Ference BA, Bhatt DL, Catapano AL, et al. Association of genetic variants related to combined exposure to lower low-density lipoproteins and lower systolic blood pressure with lifetime risk of cardiovascular disease. JAMA. 2019;322(14):1381-1391. doi:10.1001/jama.2019.14120